Mariano Aníbal Giorgi ID
Principal investigator. Health Economics and Health Technologies Unit, Instituto
Universitario CEMIC (IUC).
Associate Professor of Pharmacology,
Instituto Universitario CEMIC (IUC).
Head of Pharmacology, Construir
Salud.
Ciudad Autónoma de Buenos Aires, Argentina.
Acta Gastroenterol Latinoam 2023;53(2):109-112
Received: 20/06/2023 / Accepted: 28/06/2023 / Published online 30/06/2023 / https://doi.org/10.52787/agl.v53i2.332
Health economic evaluations represent a technical approach that provides information in the decision-making process to incorporate or not a drug, a device or a diagnostic method in a healthcare system. However, before describing them, we will review the conceptual framework that contains them.
Health and economy are closely related. As the experience of the COVID-19 pandemic has shown us, a pathological condition can disrupt the economic system of a society -of the world, in this case-. In turn, the reorganization of this system has made it possible to meet the multiple challenges of the disease: direct investment in the healthcare system, vaccines, subsidies, among others.1 Likewise, the economic conditions in which a society develops can influence the inhabitants, generate less healthy lifestyles, reduce accessibility to healthcare services and, consequently, lead to a higher prevalence and incidence of chronic non-communicable diseases (cardiovascular, oncological) and changes in mortality patterns.2
Health is recognized as a human right by the World Health Organization (WHO), which implies the a "legal obligation of States to ensure access to healthcare in a timely, acceptable and affordable way", and is part of the 2030 agenda for Sustainable Development and Universal Health Coverage.3-4 The term “affordable” implies that from the world's highest level of health planning, the WHO, takes into consideration that economic aspects are an essential part of health. All resources involved in the provision of healthcare services (called health technologies - HT) have an economic impact for beneficiaries, healthcare personnel, providers, payors, the pharmaceutical and device industry, and health authorities. Every time a person requires care or is part of a preventive intervention, there is a cost involved that has to vary depending on which part of the process we consider. Let's take a very concise example: a person who attends an outpatient consultation at a health center may have to cover transportation, loss of earnings or even some type of co-payment. On the other hand, the health center will pay for the personnel, supplies, and infrastructure. Finally, if a drug has been prescribed, the cost may be shared among the patient, the pharmacy and the funder (health insurance, the State).5 Every time we face the need to prescribe a drug, a diagnostic study, or a procedure, we must keep in mind that someone, at some point, will have to bear the cost of what we have been told to do. Therefore, it is important to be able to establish which health resources provide the greatest benefit to patients and justify the cost.
In Health Economics courses it is common to mention a quote from Antonio Machado (Proverbios y Cantares): "A fool confuses value with price". Technically speaking, we define value as the clinical benefit of a HT and price as its financial consequence.
The decision on the value (understood as clinical benefit) provided by a HT should be supported by standardized criteria based on a robust system of evidence-quality analysis.6 Systems such as GRADE (Grading of Recommendations Assessment, Development and Evaluation)7 or Levels of Evidence from the CEBM (Center for Evidence Based Medicine of the University of Oxford)8 are two of the main conceptual frameworks that have made it possible to define the quality of the evidence for different HT. In the case of GRADE, the process begins by analyzing the medical literature to find what is important to patients and then incorporating other perspectives, such as the efficacy of the interventions and the population impact that they may have, using strict criteria on the methodological quality of the sources of evidence. In the case of the CEBM, the focus is on determining which type of research design is the most appropriate to answer a question, from efficacy and safety to epidemiological and economic aspects. Although the main source of evidence on the efficacy and safety of HT comes from randomized clinical trials and meta-analyses, the results cannot be directly transferred to patients usually because of strict inclusion and exclusion criteria. To complement these sources, in recent years, real-life studies (called Real-World Evidence) has been added to provide information on the performance of a HT in real patients and healthcare scenarios.9 The other source of evidence recently incorporated into the value analysis of a HT is the patient-reported outcomes (PROs), which include the patient's perspective through studies that directly measure clinical results, for example, through scales.10 The integration of other perspectives works well in scenarios where decisions about the evidence are not easy. For example, a new drug may provide an increase in overall survival but at the cost of a high incidence of serious adverse events and, consequently, imply a poor quality of life for patients. Therefore, the clinical benefit can be measured in terms of events (for example, mortality, hospitalizations) or by using measures that combine survival and quality of life such as Quality Adjusted Life Years (QALY).11 Finally, when considering the clinical benefit of HT, it is important to highlight that each country or jurisdiction has its own characteristics (population, infrastructure, human resources, accessibility to the healthcare system, access to drugs) that could also modify the clinical results of a disease12 and the expected impact of HT on specific diseases or conditions.13
Regarding the price (or cost, depending on your point of view), it is important to note that not only the purchase or acquisition price of a HT should be considered, but also all the costs associated with disease management, as previously indicated.5 In other words, a HT may have a high acquisition cost but represents a lower final cost because it results in a lower use of resources (for example, hospitalizations, deaths). The cost is different for the various participants in the healthcare system and varies according to the type of HT (an innovative therapy will be more expensive, for instance), the duration of use (for example, lifelong treatments) and, most importantly, the country we are in. Therefore, costs cannot be extrapolated or applied from one jurisdiction to another, which is technically known as non-transferability.14
This is undoubtedly one of the most difficult questions to answer. Two central elements must be considered to face this dilemma: 1) the value and quality of the evidence provided by a HT and 2) affordability. A globally used approach for many decades has been the value-based pricing. Briefly, it is about establishing an acceptable price for a health benefit, for example, price per QALY gained. In some countries this price is well defined and represents the expense that can be made to incorporate a new HT considering equity in the use of resources.15 For example, the UK typically places this price between £ 20,000 and £ 30,000 per QALY gained. In other countries, this threshold is defined using gross domestic product (GDP) per capita which intends to represent the ammount of money that would be appropriate to invest in order to improve healthcare for the population.16 These thresholds define what a healthcare system is willing to pay to achieve a health benefit and to claim that a HT is cost-effective.
Economic evaluations are a set of analytical methods that provide information on the economic impact of a HT. One branch of health economic evaluations deals with drugs use and is known as pharmacoeconomics (PE). The methodology of pharmacoeconomic analysis is standardized, allowing methodological robustness. One of the main sources of standardization comes from the International Society for Pharmacoeconomics and Outcomes Research (ISPOR), which publishes consensus for conducting PE studies.17 Basically, PE studies compare two or more therapeutic options for a given disease or condition. They measure clinical costs and benefits for all options using the same criteria.
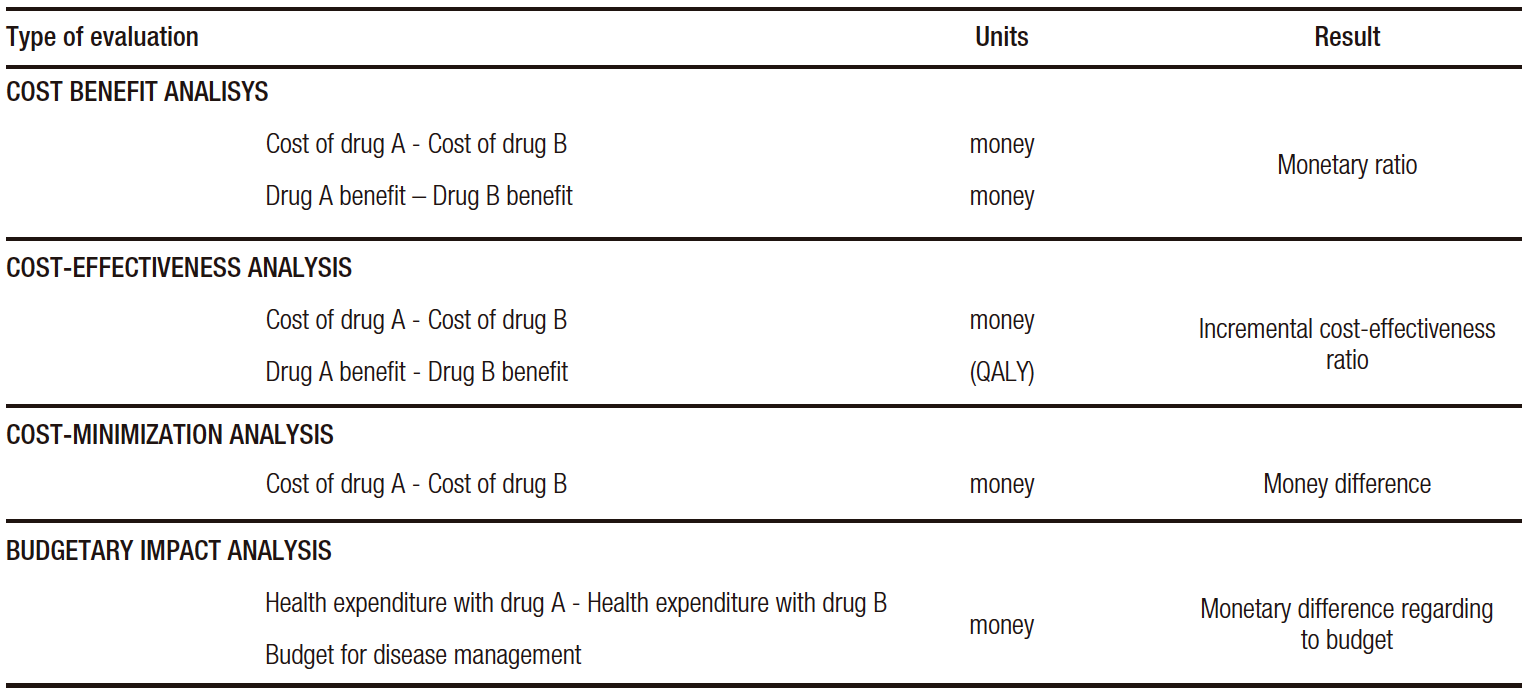
The basic types of analysis most commonly used in pharmacoeconomics are: cost-benefit, cost-effectiveness, cost-minimization and budgetary impact (Table 1).
Table 1. Main types of health economic evaluations
Cost-Benefit Analysis (CBA): this type of study measures both health costs and benefits in monetary units (US dollars, for example). This approach contemplates only the economic value of health. CBAs are very useful in order to assess the financial impact of health programs that involve pharmaceutical interventions.
Cost-Effectiveness Analysis (CEA): strictly speaking, in this type of study, costs are measured in monetary units and health outcomes in units of clinical effectiveness as life years gained, deaths or events avoided, and in QALY. If we compare two drugs, A and B, for each drug, there would be a clinical effectiveness measure (per QALY) and the total cost of using each. Thus, there would be a cost-benefit ratio for each of them. The difference between the two is the incremental cost-effectiveness ratio (ICER), which expresses how much more is paid for the extra health benefit obtained. This measure is correlated with payment thresholds mentioned previously.
Cost-Minimization Analysis (CMA): this analysis compares the costs of two drugs that have the same clinical effectiveness. It is a simple form of evaluation that allows a quick approach to the problem.
Budgetary Impact Analysis (BIA): this analysis examines the overall healthcare expenditure involved in the use of one drug compared to another in relation to the health budget. It contemplates the fact that healthcare resources have a defined limit.
Although each of these tools has specific technical characteristics, they share the fact that they integrate value and price into a single evaluation and ultimately provide a standardized measurement.
Without being specialists in the field, healthcare physicians are increasingly exposed to studies of these characteristics. Although we have mentioned that there are standardized methods, it is important to highlight some considerations that we must keep in mind to assess the scientific literature in this field:
1. Quality of evidence: the study should clearly report which studies were considered to estimate the clinical effectiveness of HT.
2. Quality of cost data: although it is not possible to precisely know the health costs of each system or jurisdiction, at least the sources of these costs should be reported.
3. Local data: the economic evaluation should be carried out or adapted to the country or jurisdiction where the use of HT is being considered. That is, it should have epidemiological information, resource use data, and local cost information.
Finally, considering that the current economic conditions may be unstable, the studies should also provide information on this point.18
Intellectual Property. The author declares that the data and table presented in the manuscript are original and were carried out at his belonging institution.
Funding. The author declares that there were no external sources of funding.
Conflict of interest. The author declares that he has no conflicts of interest in relation to this article.
Copyright © 2023 Acta Gastroenterológica latinoamericana. This is an
open-access article released under the terms of the Creative Commons Attribution (CC BY-NC-SA 4.0) license,
which allows non-commercial use, distribution, and reproduction, provided the original author and source are
acknowledged.
© 2023 Acta Gastroenterológica latinoamericana. This is an
open-access article released under the terms of the Creative Commons Attribution (CC BY-NC-SA 4.0) license,
which allows non-commercial use, distribution, and reproduction, provided the original author and source are
acknowledged.
Cite this article as: Giorgi M A. Health Economic Evaluations: what should we take into account when reading them?. Acta Gastroenterol Latinoam. 2023;53(2):109-112. https://doi.org/10.52787/agl.v53i2.332
Correspondence: Mariano Aníbal Giorgi
Email: marianoagiorgi@gmail.com
Acta Gastroenterol Latinoam 2023;53(2):109-112