Manuel Barbero ID· Ignacio Roca ID· Lucía Navarro ID· Nicolás Domínguez ID· Omar Galdame ID· María Del Carmen Puente ID· Laura González ID· Lucía Guillén ID· María Virginia Gustincic ID· Fernando Cairo ID
Hospital El Cruce.
Florencio Varela, Provincia de Buenos Aires. Argentina
Acta Gastroenterol Latinoam 2026;56(1):71-77
Received: 20/01/2026 / Accepted: 19/03/2026 / Published online: 31/03/2026 / https://doi.org/10.52787/agl.v56i1.595
Introduction. Anti-HCV seropositivity is a public-health concern in Argentina, with potential disparities by gender. We estimated anti-HCV seropositivity in a multicenter public laboratory network, described gender-based differences, and examined the association with all-cause mortality. Methods. Retrospective observational study with centralized chemiluminescent immunoassay testing at Hospital El Cruce, using samples from 64 public centers from January 1, 2013 to December 31, 2023. The analyses were performed at the patient level (the first test per person) and the positivity rate was calculated for each testing site. Vital status information was obtained from hospital records and RENAPER (with a cut-off date of December 2024). Mortality by anti-HCV serostatus was assessed using logistic regression with a prespecified 2:1 matching on age, gender, and testing site. Results. Of the 74,503 adults tested, 1,101 (1.48%) were anti-HCV–positive. Of those tested, 83.0% were women (61,837/74,503) and 17.0% were men (12,666/74,503). Within gender seropositivity was 0.73% in women (451/61,837) and 5.05% in men (639/12,666). The per-test positivity rate was 0.5% in peripheral centers (451/90,194) and 5.1% in the hospital (922/18,082). Of the individuals who tested positive for anti-HCV, 25.0% (275/1,101) were deceased at the cut-off date. In a matched cohort of 2:1 by age, gender, and site, anti-HCV positivity was associated with a higher risk of death (adjusted OR 4.75; 95% CI 3.82 – 5.94; p < 0.001). Conclusions. Men were tested much less frequently than women (17.0% versus 83.0% of those tested), yet they were far more likely to test positive for anti-HCV (5.05% versus 0.73% within gender), accounting for the majority of positive results (58%). Therefore, screening policies should prioritize increasing testing among men, especially in high-yield hospital settings, and ensure rapid linkage to care. The association between anti-HCV seropositivity and higher mortality reinforces the urgency of these actions.
Keywords. Hepatitis C/epidemiology, serologic tests, mass screening, sex factors, mortality, Argentina/epidemiology.
Introducción. La seropositividad al anti-VHC es un problema de salud pública en Argentina, con posibles disparidades según el género. Estimamos la seropositividad al anti-VHC en una red multicéntrica de laboratorios públicos, describimos las diferencias según el género y examinamos la asociación con la mortalidad por todas las causas. Métodos. Se realizó un estudio observacional retrospectivo con pruebas centralizadas de inmunoensayo quimioluminiscente en el Hospital El Cruce, utilizando muestras de 64 centros públicos, desde el 1° de enero de 2013 al 31 de diciembre de 2023. Los análisis se realizaron a nivel de paciente (primera prueba por persona); se calculó la positividad por centro de análisis. La información sobre el estado vital se obtuvo de los registros hospitalarios y del RENAPER (fecha límite: diciembre de 2024). Se evaluó la mortalidad según el estado serológico anti-VHC mediante regresión logística, con un emparejamiento pre-especificado 2:1 por edad, género y centro de análisis. Resultados. De los 74.503 adultos sometidos a pruebas, 1.101 (1,48%) fueron anti-VHC positivos. De los pacientes estudiados, 83,0% fueron mujeres (61.837/74.503) y 17,0% hombres (12.666/74.503). Según el sexo, la seropositividad fue 0,73% entre las mujeres (451/61.837) y 5,05% entre los hombres (639/12.666). La positividad por prueba fue 0,5% en los centros periféricos (451/90.194) y 5,1% en el hospital (922/18.082). Entre las personas anti-VHC positivas, el 25,0% (275/1.101) había fallecido a la fecha de corte. En una cohorte emparejada 2:1 por edad, sexo y centro, la positividad al anti-VHC se asoció con un riesgo mayor de muerte (OR ajustado 4,75; IC del 95%: 3,82–5,94; p < 0,001). Conclusiones. Los varones fueron estudiados con menor frecuencia que las mujeres (17.0% versus 83.0% de los testeados), aunque tenían una probabilidad mayor de ser positivos para el anti-VHC (5.05% versus 0.73% según el género). Las políticas de tamizaje deberían priorizar el incremento de las pruebas entre los hombres, especialmente en los entornos hospitalarios de alto rendimiento, garantizando una vinculación rápida con la asistencia del paciente. La asociación entre la seropositividad del anti-HCV y una mayor mortalidad refuerza la urgencia de estas acciones.
Palabras claves. Hepatitis C/epidemiología, pruebas serológicas, tamizaje masivo, factores relacionados con el sexo, mortalidad, Argentina/epidemiología.
Abbreviation
HCV: Hepatitis C virus.
CLIA: Chemiluminescent immunoassay.
S/CO:
Signal-to-cutoff ratio.
RNA: Ribonucleic acid.
RENAPER: Registro Nacional de las
Personas (Argentina’s National Registry of Persons).
SD: Standard
deviation.
IQR: Interquartile range.
CI: Confidence interval.
χ²:
Chi-square.
OR: Odds ratio.
US: United States.
Hepatitis C virus (HCV) infection is a significant public health concern, with substantial implications for morbidity and mortality worldwide. A key step in achieving the World Health Organization’s goal of eliminating hepatitis C by 2030 is to identify individuals at risk using robust screening methods to detect undiagnosed cases.1 However, many countries, including Argentina, face challenges in conducting effective hepatitis C screening and surveillance.2-3
While the global prevalence of hepatitis C virus infection is estimated to be around 2.5%, there are significant geographic variations in incidence and prevalence.4 HCV infection remains a major public health challenge in Argentina, being a leading cause of cirrhosis, hepatocellular carcinoma, liver transplantation, and liver-related mortality.5 Despite advances in treatment, a significant proportion of infected individuals remain undiagnosed and untreated, contributing to the ongoing burden of HCV-related complications.
In Argentina, available data suggest that hepatitis C prevalence may range from 0.7% to 2% of the population.5 Furthermore, anti-HCV positivity has been associated with increased overall mortality.7-9
Globally, women utilize healthcare services more frequently than men, largely due to routine gynecological checkups, prenatal care, and childbirth.10 This increased interaction with the healthcare system may contribute to higher screening rates for various conditions, including infectious diseases such as hepatitis C.
The true prevalence of hepatitis C in Argentina is uncertain due to a lack of comprehensive epidemiological studies. This lack of data makes it difficult to assess the effectiveness of current screening strategies and to fully understand potential gender disparities in anti-HCV seropositivity.5 The main difficulties in obtaining demographic data are related to Argentina’s sociocultural, geographic, and economic aspects.
The aim of this study is:
1. To determine anti-HCV seropositivity in a multicenter public laboratory network in Argentina.
2. To investigate gender-based differences in the proportion tested and anti-HCV seropositivity.
3. To determine the association between anti-HCV seropositivity measured by CLIA and all-cause mortality.
The aim of these findings is to inform public health policies by identifying critical gaps in gender-based screening and positivity patterns.
Study design and setting
We conducted a multicenter retrospective observational study using data from the ReLink program in Argentina between 1 January 2013 and 31 December 2023. Blood samples were collected at 64 low–medium complexity public health centers and at a high-complexity referral hospital (Hospital El Cruce, Buenos Aires), where all serological analyses were centralized. Positivity by testing site was computed per test; all other estimates were calculated at the patient level (first test per person).
Participants
We included adults (≥ 18 years) who had at least one anti-HCV test during the study period. For individuals with multiple tests, analyses were performed at the individual level using the first test as the reference point. Records were de-duplicated using a unique identifier. No additional exclusion criteria were applied.
Data sources and measurements
Demographic and testing data were extracted from the central laboratory information system. Serum/plasma samples were tested for anti-HCV using a chemiluminescent immunoassay (CLIA). The results followed the manufacturer’s cut-off: reactive if the signal-to-cutoff (S/CO) ratio was ≥ 1.0 and non-reactive if S/CO ratio was < 1.0. Initially reactive results were retested, and those that remained reactive were classified as anti-HCV positive. Reflex HCV-RNA confirmation was only performed among individuals who were re-linked to care within the ReLink program, and this is reported elsewhere. RNA was not used to define seropositivity in the present analysis. The testing site was classified as hospital (Hospital El Cruce) or peripheral center (the 64 public centers). Vital status (all-cause mortality) was ascertained by record linkage with hospital records and the RENAPER national registry, with a cut-off date of December 2024.
Outcomes
The primary outcomes were: (1) anti-HCV seropositivity (yes/no) and (2) all-cause mortality at the mortality cut-off.
Bias and study size
The study included all eligible tests during the specified period (census of available testing). Potential biases (selection, misclassification and residual confounding) were mitigated through centralized testing and de-duplication at person level.
Statistical analysis
Continuous variables are reported as mean (and SD ifavailable) or median (IQR) following a distribution assessment using the Shapiro–Wilk test. Categorical variables are shown as counts (%), and 95% confidence intervals (CIs) are reported for comparative estimates when applicable. Group comparisons used a t-test or Mann–Whitney U test for continuous variables and χ² (or Fisher’s exact test when expected counts < 5) for categorical variables. To evaluate the association between HCV serostatus and mortality, we fitted a logistic regression model, using anti-HCV–positive vs. anti-HCV–negative as the exposure and all-cause mortality as the outcome, adjusting for age (continuous), gender, and testing site (hospital vs. peripheral center). We assessed linearity in the logit for age and checked for multicollinearity. Additionally, we conducted a secondary matched analysis using a 2:1 ratio for gender, age, and testing site, and fitted a logistic regression model within the matched cohort. The results are reported as odds ratios (OR) with 95% CI. Statistical significance was defined as p < 0.05. There were no missing values for the core variables (age, gender, testing site) so analyses were complete-case without imputation.
Ethics
The study was approved by the Ethics Committee at Hospital El Cruce. As it was a retrospective study using de-identified data, informed consent was waived.
Software
Analyses were performed in R, version 4.2.1.
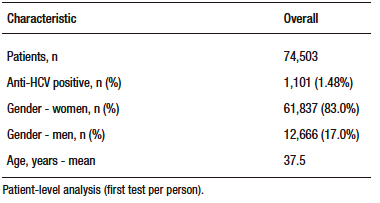
A total of 108,276 samples were processed using chemiluminescence, corresponding to 74,503 unique patients. Of these, 1,680 samples were reactive, belonging to 1,101 individuals, giving an overall anti-HCV seropositivity rate of 1.48% (1,101/74,503). Analyses were performed at the individual level using the first test result for each patient; there were no missing data for age, gender, or testing site. Follow-up for mortality extended through to December 2024 (Table 1).
Tabla 1. Baseline characteristics of patients tested for HCV (patient-level)
Most tests originated from peripheral public health centers, with a smaller proportion originating from the hospital setting. At the test level, peripheral centers accounted for 90,194 tests (83.3%), while the hospital accounted for 18,082 tests (16.7%). Anti-HCV seropositivity per test was 0.5% (451/90,194) in peripheral centers and 5.1% (922/18,082) in hospital settings (Figure 2).
Of those tested, 83% were women (61,837) and 17% were men (12,666). The proportion
of women who were seropositive was 0.73% (451/61,837) and the proportion of men were 5.05% (639/12,666)
(Figure 1).
The mean age of the tested population was 37.5 years (SD ± 16) years, with women at
35.5 years (SD ± 14) and men at 45.3 years (SD ± 20). Of the 1,101 individuals who tested positive for HCV, 451
were women (40.9%) and 639 were men (58.0%). The mean age of HCV-positive participants was 48.3 ± 14 years
overall, 44.6 ± 16 years in women and 50.7 ± 10 years in men.
Figure 1. Seropositivity by gender (patient-level)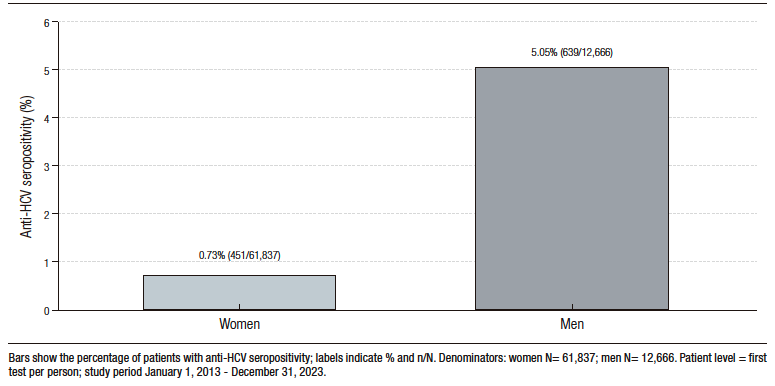
Figure 2. Seropositivity per test by testing site, 2013-2023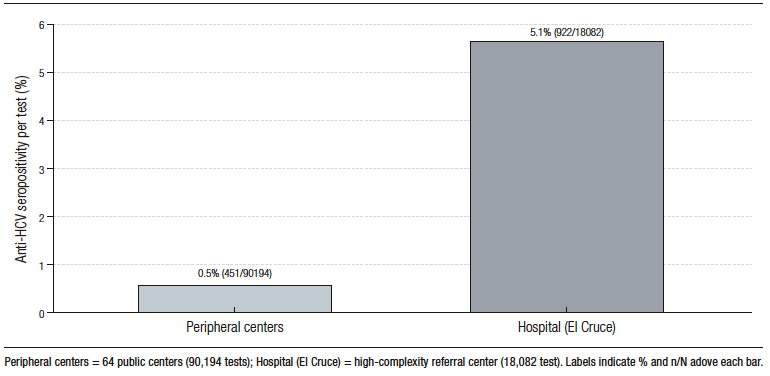
Regarding mortality, 275/1,101 (25.0%) anti-HCV–positive patients had died by the December 2024 cut-off. In a logistic regression fitted on a cohort matched 2:1 by gender, age, and testing site, anti-HCV positivity was associated with substantially higher odds of death versus anti-HCV negativity (adjusted OR 4.75; 95% CI 3.82–5.94; p < 0.001) (Table 2).
Tabla 2. All-cause mortality by anti-HCV serostatus (patient-level; matched 2:1
cohort; cut-off Dec 2024)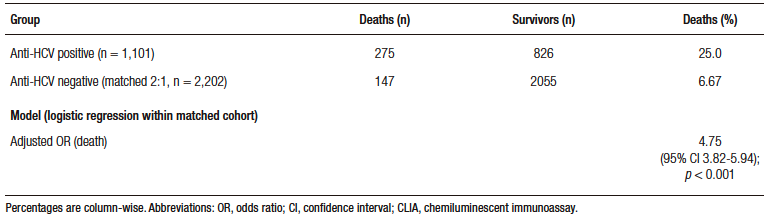
Our study highlights the significant burden of hepatitis C in Argentina, with an anti-HCV seropositivity rate of 1.48%, which is consistent with national prevalence estimates ranging from 0.7% to 2%.5 These findings are consistent with those of a population-based study conducted by Mendizabal et al. in Buenos Aires Province.11 We also incorporated data from a recent national study of blood donors, which reported an anti-HCv seroreactivity rate of 0.79% between 2017 and 2021. This confirms the low but persistent prevalence of HCV in the general healthy population.12 In addition, our results corroborate prior surveillance data from Flichman et al., which suggests that hospital-based testing identifies a higher proportion of positives cases.13
Although women represented the majority of individuals tested, likely due to their higher engagement with the healthcare system, (including routine gynecologic visits and pregnancy-related care), men had a substantially higher anti-HCV positivity rate.14 Despite being underrepresented in the screened population, men accounted for 58% of anti-HCV–positive cases. This suggests either delayed diagnosis or a higher cumulative risk, underscoring the need for gender-targeted screening strategies. These patterns are consistent with a national report and echo findings from studies in other Latin American settings, such as Cobo et al., who described how gender and structural inequalities affect healthcare access.15, 16
The observed gender disparity may also reflect differences in underlying risk factors, such as intravenous drug use, occupational exposures, or sexual behaviors.17 Although our study did not evaluate these directly, the findings support the need to further explore gender based vulnerability within the Argentine context.
Argentina, like many Latin American countries, faces structural barriers to hepatitis C elimination, including socioeconomic disparities and gaps in access to testing and treatment. Understanding the local epidemiology is essential for designing effective public health strategies.13
We also observed a strong association between anti-HCV positivity and mortality. At the time of analysis, 25% of anti-HCV–positive individuals had died, and anti-HCV positivity was associated with a nearly five-fold increase in the odds of mortality (OR 4.75; 95% CI 3.82-5.94; p < 0.001), compared to a matched cohort of anti-HCV–negative individuals. This finding is consistent with previous reports by Omland et al. and Neal et al., which documented increased mortality in HCV-infected cohorts.8, 9 Similar results were reported by El-Kamary et al. in the US, where all-cause mortality was more than doubled in individuals with chronic HCV.18 It is important to note that our mortality data, derived from RENAPER, reflect all-cause mortality only. We were unable to determine the specific cause of death for each individual. Therefore, we cannot quantify the proportion of deaths attributable to hepatic causes (such as cirrhosis, hepatocellular carcinoma, or liver failure) versus non-hepatic causes (such as cardiovascular disease, infectious complications, or other comorbidities). Despite this limitation, the magnitude of the observed excess mortality in anti-HCV–positive individuals highlights the clinical and public health importance of HCV infection beyond liver disease alone.
These results emphasize the critical importance of early detection, prompt treatment initiation, and robust re-linkage programs for those who test positive. In a context where many remain undiagnosed, central laboratories can play a key role in identifying and referring high-risk individuals for timely care.
Strengths and Limitations
The strengths include the large, multicenter sample drawn from 64 public centers and a high-complexity referral hospital, centralized chemiluminescent serological testing with uniform cut-offs, deterministic de-duplication at the individual level, and linkage to hospital records and RENAPER for vital status. Limitations include the lack of comprehensive nationwide sampling, reliance on a single laboratory network, the retrospective design (where testing was performed for clinical indications, limiting generalizability to the general population and precluding inference of national prevalence) and the absence of cause-specific mortality. Vital status was ascertained through record linkage with RENAPER (Argentina’s National Registry of Persons), which provides reliable all-cause mortality data but does not include information on the underlying cause of death. This precludes any inference about the proportion of deaths that are directly attributable to HCV-related liver disease versus those that occurr due to comorbid or unrelated conditions. Future studies with access to death certificate data or clinical records are needed to characterize cause-specific mortality in this population. A prospective study with a broader and more representative population is required to validate these conclusions.
In conclusion, this study provides insights into the epidemiology of hepatitis C in Argentina, highlighting the importance of strengthening screening and surveillance efforts to address potential gender-based disparities. Targeted interventions to enhance HCV detection and linkage to care are crucial in reducing the disease’s burden and improving public health outcomes. The demonstrated association between anti-HCV positivity and a higher risk of death emphasizes the importance of implementing effective strategies for diagnosis, care linkage, and treatment initiation, with the aim of mitigating the long-term consequences of chronic HCV infection in Argentina.
Consent for publication. Written informed consent was obtained from the patient for participation and publication of this case.
Intellectual property. The authors declare that the data, figures and tables in this article are original and were carried out at their institutions.
Funding. This study was supported by a research grant from Gilead Sciences.
Conflict of interest. The authors declare that they have no conflicts of interest in relation to this article.
Copyright

© 2026 Acta Gastroenterológica latinoamericana. This is an open-access article released under the terms of the Creative Commons Attribution (CC BY-NC-SA 4.0) license, which allows non-commercial use, distribution, and reproduction, provided the original author and source are acknowledged.
Cite this article as: Barbero M, Roca I, Navarro L et al. Hepatitis C Screening and Gender Disparities in Positivity Rates in Argentina: A Multicenter Study. Acta Gastroenterol Latinoam. 2026;56(1):71-77. https://doi.org/10.52787/agl.v56i1.595
Correspondence: Manuel Barbero
Email: manuelbarbero11@gmail.com
Acta Gastroenterol Latinoam 2026;56(1):71-77