Juan I. Olmos1 ID· John E. Pandolfino2 ID
1Esophageal and Neurogastroenterology Section. Division of Gastroenterology and
Hepatology. Hospital de Clínicas “José de San Martín”, University of Buenos Aires.
Argentina.
2Kenneth C. Griffin Esophageal Center, Division of Gastroenterology and
Hepatology Feinberg School of Medicine, Northwestern University, Chicago, IL, USA.
Acta Gastroenterol Latinoam 2026;56(1):29-40
Received: 01/02/2026 / Accepted: 06/03/2026 / Published online: 31/03/2026 / https://doi.org/10.52787/agl.v56i1.601
Esophageal symptoms -including dysphagia, heartburn, regurgitation, and non-cardiogenic chest pain- are among the most common indications for referral to gastroenterology. Despite their high prevalence, establishing a definitive etiological diagnosis remains a significant clinical challenge due to the marked overlap in symptom profiles between gastroesophageal reflux disease, eosinophilic esophagitis, esophageal motility disorders, and disorders of gut–brain interaction. Current diagnostic paradigms are generally based on sequential testing strategies that often begin with empirical treatment with proton pump inhibitor followed by endoscopy and, when this is inconclusive, catheter-based esophageal functional studies. This approach is associated with prolonged diagnostic times, incomplete studies due to poor tolerance, delays in access to specialized centers, a substantial increase in healthcare costs. Recent advances in endoscopic and physiologic technologies offer the opportunity to fundamentally reframe the evaluation of esophageal symptoms. The integration of endoscopic risk stratification tools, such as the Clinical Achalasia Risk Score, Functional Lumen Imaging Probe panometry, and wireless pH monitoring, enables a comprehensive physiologic evaluation during the index endoscopy under sedation, optimizing the diagnostic performance of the procedure. This strategy has the potential to significantly reduce diagnostic uncertainty, shorten the time to definitive diagnosis, and decrease reliance on poorly tolerated catheter studies. This review synthesizes the emerging evidence supporting a diagnostic paradigm centered on index endoscopy as a central and integrative diagnostic event. The limitations of traditional diagnostic methods are analyzed and the physiological principles and clinical utility of planimetry using Functional Lumen Imaging Probe and wireless reflux monitoring are reviewed. A practical algorithm integrating these tools is proposed, with the aim of providing accurate and efficient care, aimed at improving diagnostic performance and therapeutic decision-making in patients with esophageal symptoms. Given that the available evidence is still emerging and concentrated in centers with experience, the proposed algorithm should be interpreted as a pragmatic framework for optimizing the diagnostic sequence, subject to prospective validation and adaptation to the local context.
Keywords. Index endoscopy, FLIP planimetry, esophageal motility disorders, wireless pH monitoring, Clinical Achalasia Risk Score, gastroesophageal reflux disease, eosinophilic esophagitis, esophagogastric junction, high-resolution manometry, dysphagia.
Los síntomas esofágicos -incluyendo la disfagia, la pirosis, la regurgitación y el dolor torácico no cardiogénico- se encuentran entre las indicaciones más frecuentes de derivación a gastroenterología. A pesar de su elevada prevalencia, el establecimiento de un diagnóstico etiológico definitivo continúa siendo un desafío clínico relevante, debido a la marcada superposición de perfiles sintomáticos entre la enfermedad por reflujo gastroesofágico, la esofagitis eosinofílica, los trastornos motores esofágicos y los trastornos de la interacción intestino-cerebro. Los paradigmas diagnósticos vigentes se basan, en general, en estrategias secuenciales que con frecuencia comienzan con un tratamiento empírico con inhibidores de la bomba de protones, seguido de una endoscopía digestiva alta y, cuando esta no resulta concluyente, de estudios funcionales esofágicos basados en catéter. Este enfoque se asocia a tiempos diagnósticos prolongados, estudios incompletos por mala tolerancia, demoras en el acceso a centros especializados y un incremento sustancial de los costos sanitarios. Los avances recientes en las tecnologías endoscópicas y funcionales ofrecen la oportunidad de replantear de manera fundamental la evaluación de los síntomas esofágicos. La integración de herramientas de estratificación endoscópica de riesgo -como el Clinical Achalasia Risk Score- junto con la planimetría mediante Functional Lumen Imaging Probe y el monitoreo inalámbrico del pH, permite realizar una evaluación fisiológica integral durante la endoscopía índice, bajo sedación, optimizando el rendimiento diagnóstico del procedimiento. Esta estrategia tiene el potencial de reducir significativamente la incertidumbre diagnóstica, acortar el tiempo hasta el diagnóstico definitivo y disminuir la dependencia de estudios con catéter mal tolerados. En esta revisión se sintetiza la evidencia emergente que respalda un paradigma diagnóstico centrado en la endoscopía índice como evento diagnóstico central e integrador. Se analizan las limitaciones de los métodos diagnósticos tradicionales, se revisan los principios fisiológicos y la utilidad clínica de la planimetría mediante Functional Lumen Imaging Probe y el monitoreo inalámbrico de reflujo. Se propone un algoritmo práctico que integra esas herramientas, con el objetivo de brindar una atención de precisión y eficiente, orientada a mejorar el rendimiento diagnóstico y la toma de decisiones terapéuticas en los pacientes con síntomas esofágicos. Dado que la evidencia disponible es emergente y se concentra en centros con experiencia, el algoritmo propuesto debe interpretarse como un marco pragmático para optimizar la secuencia diagnóstica, sujeto a validación prospectiva y adaptación al contexto local.
Palabras claves. Endoscopía índice, planimetría FLIP, trastornos motores esofágicos, monitoreo inalámbrico del pH, Clinical Achalasia Risk Score, enfermedad por reflujo gastroesofágico, esofagitis eosinofílica, unión esofagogástrica, manometría de alta resolución, disfagia.
Abbreviation
GERD: Gastroesophageal reflux disease.
EoE: Eosinophilic esophagitis.
PPIs:
Proton pump inhibitors.
CARS: Clinical Achalasia Risk Score.
FLIP: Functional Lumen
Imaging Probe.
pH: Hydrogen potential.
HRM: High-resolution
manometry.
EGJ: Esophagogastric junction.
EREFS: Edema, rings, exudates, furrows,
strictures.
IRP: Integrated relaxation pressure.
EGJ-DI: Esophagogastric junction
distensibility index.
RAC: Repetitive anterograde contractions.
AET: Acid exposure
time.
Esophageal symptoms, such as heartburn, dysphagia, regurgitation, chest pain, and food impaction, account for a substantial proportion of outpatient gastroenterology encounters. Although these are common clinical manifestations, the underlying etiologies are heterogeneous and, in many cases, difficult to differentiate based solely solely on clinical history. Gastroesophageal reflux disease (GERD), eosinophilic esophagitis (EoE), esophageal motility disorders, and functional esophageal disorders often present with overlapping symptom profiles. This leads to diagnostic ambiguity and therapeutic uncertainty.1
Historically, the evaluation of esophageal symptoms has relied on empiric therapeutic trials, particularly with proton pump inhibitors (PPIs), followed by upper endoscopy and, when necessary, esophageal function testing. Although this model is firmly established in routine clinical practice, there is a growing recognition that this approach may be diagnostically inefficient and ineffective in establishing the underlying etiology of symptoms in a significant number of cases. Empirical treatment is associated with a significant placebo effect, delays in definitive diagnosis, and unnecessary prolongation of patient discomfort, particularly in those with esophageal motility disorders or gut-brain interaction disorders.2, 3
Endoscopy remains a cornerstone of esophageal evaluation due to its ability to rule out malignancy, identify mucosal disease, and detect mechanical obstruction. However, up to 50 - 70% of patients undergoing upper endoscopy for esophageal symptoms show no obvious abnormalities, leaving clinicians with limited diagnostic clarity and patients with persistent symptoms.3 In these cases, esophageal functional tests -high-resolution manometry (HRM), ambulatory reflux monitoring, and barium esophagram-are necessary to establish a diagnosis. Unfortunately, these tests are often performed weeks or months later, require referral to specialized centers, and are limited by suboptimal tolerance and a significant proportion of incomplete studies.2, 3
Advances in endoscopic physiologic assessment now allow us to go beyond the purely exclusionary role of organic pathology in endoscopy. During endoscopy under sedation, tools such as FLIP planimetry and wireless pH monitoring (capsule placement) can be used, providing real-time assessment of esophageal motility, esophagogastric junction (EGJ) opening dynamics, and reflux burden. When combined with structured endoscopic risk stratification systems, such as the Clinical Achalasia Risk Score (CARS),4, 5 these technologies enable an integrated precision diagnostic approach during index endoscopy.
In this context, this review proposes a paradigm shift in the evaluation of esophageal symptoms, positioning index endoscopy as a central and integrative diagnostic event rather than a preliminary screening test. The integration of endoscopic evaluation, physiological studies, and objective reflux monitoring in a single study reduce diagnostic delays, improve the patient experience, and optimize diagnostic and therapeutic decision-making. It should be noted that this strategy is supported by recent but still expanding evidence, so its implementation requires consideration of technological availability, team training, and specific clinical scenarios.
Limitations of the current diagnostic paradigm
Prolonged diagnostic timelines
The traditional diagnostic approach to evaluating esophageal symptoms is inherently sequential and time-consuming. Patients with heartburn or chest pain are often treated empirically with PPIs for 4 - 8 weeks before further evaluation is considered. When symptoms persist, an upper endoscopy is performed, and ambulatory reflux monitoring or manometry studies are often indicated if endoscopic findings are inconclusive. This stepwise approach commonly results in diagnostic times ranging from three to six months, and in some cases exceeding one year.2
Dysphagia is a particularly illustrative example of these limitations. Although endoscopy is appropriately prioritized for its role in ruling out malignant pathology, a normal endoscopic examination frequently leads to the need for additional evaluations. Achalasia and other motility disorders are especially susceptible to diagnostic delays, as the early stages of the disease may present with normal endoscopic findings or equivocal imaging studies. Several studies have shown that patients with achalasia often experience diagnostic delays of more than 12 months, during which time symptoms are often mistakenly attributed to GERD or functional disorders.3
Excessive reliance on empirical treatment
Empirical treatment with proton pump inhibitors (PPIs) continues to be a widely used initial strategy in patients with esophageal symptoms, particularly heartburn and chest pain. While this approach may be appropriate in selected populations, its indiscriminate use poses significant limitations from a diagnostic standpoint. The clinical response to PPIs does not allow for reliable differentiation between gastroesophageal reflux disease, reflux hypersensitivity, and functional heartburn. The observed symptomatic improvement may reflect a placebo effect rather than a change in the disease.6 Conversely, patients with motility disorders or EoE may experience diagnostic delays due to a transient or absent response to acid suppression.
Fragmentation of studies and barriers to access
Esophageal functional tests are essential for diagnosing motility disorders and characterizing GERD, but access to these tests is limited. High-resolution manometry and pH-impedance monitoring require specific equipment and specially trained personnel, resources that are often concentrated in tertiary centers. Wait times of several months are common, particularly in community settings. Furthermore, incomplete or failed studies occur in up to 20% of cases due to intolerance, technical difficulties, or patient anxiety.2, 3
Patient experience and test tolerability
Catheter-based diagnostics are a major barrier to effective evaluation. Transnasal manometry and pH-impedance monitoring are associated with discomfort, nausea, nasal pain, and procedural anxiety. In a prospective study evaluating patient experience with esophageal diagnostics, HRM was the only study significantly associated with a traumatic experience (χ² = 8.92, p = 0.003).7 These factors contribute to test refusal, incomplete studies, and diagnostic delays.
Rationale for optimizing index endoscopy
Given the limitations of the current paradigm, there is a strong rationale for rethinking the role of index endoscopy in the evaluation of esophageal symptoms. Endoscopy is already an inevitable endpoint for most patients due to alarm symptoms, refractory disease, or diagnostic uncertainty.1 Taking advantage of this index endoscopy to perform a physiological assessment under sedation offers multiple advantages.
First, endoscopy provides critical anatomic and mucosal information that contextualizes physiological findings. The grading of esophagitis using the Los Angeles classification, the evaluation of EoE with systematic biopsies and EREFS scoring, the assessment of the gastroesophageal valve and hiatal hernia, as well as the identification of endoscopic signs suggestive of achalasia, provide fundamental elements for the integrated interpretation of subsequent physiological data.8
Second, the introduction of structured endoscopic risk stratification tools, such as the Clinical Achalasia Risk Score (CARS), allows the probability of achalasia to be estimated based on reproducible endoscopic findings. The CARS incorporates assessment of esophageal contents, luminal anatomy, upper gastrointestinal tract resistance, and stasis-related mucosal changes, demonstrating excellent predictive value for achalasia when scores exceed predefined thresholds.4, 5
Third, planimetry using Functional Lumen Imaging Probe (FLIP) technology enables direct assessment of EGJ opening dynamics and esophageal contractile response during endoscopy under sedation. Unlike HRM, which evaluates swallow-induced peristalsis, FLIP technology assesses secondary distension-induced peristalsis as well as esophageal wall stiffness, providing particularly valuable complementary physiological information in patients with equivocal or borderline manometric findings.9-12
Finally, the wireless pH monitoring capsule can be placed during the same procedure, allowing for objective assessment of acid exposure over 48 - 96 hours without the discomfort of transnasal catheters. This approach aligns with contemporary GERD guidelines and facilitates accurate phenotyping of reflux disease early in the diagnostic course.13, 14
Taken together, these advances support the transition toward a comprehensive and precise diagnostic strategy based on index endoscopy as the central and integrative diagnostic event in the management of esophageal symptoms.
However, it should be noted that the availability of FLIP planimetry and long-term wireless pH monitoring varies across Latin America. In this context, a staged implementation of the index endoscopy approach could be considered, prioritizing its use in patients with a higher pretest probability of motility disorders or optimizing referrals to referral centers.
Endoscopic motility assessment using FLIP planimetry
Physiologic basis of FLIP planimetry
The functional lumen imaging probe (FLIP) was developed to assess the mechanical and neuromuscular properties of the esophagus using impedance planimetry during controlled volumetric distension. Unlike high-resolution manometry (HRM), which assesses the pressure patterns generated by primary peristalsis induced by swallowing, FLIP planimetry evaluates the esophageal response to sustained distension, thereby assessing secondary peristalsis and esophageal wall stiffness.
During FLIP planimetry, a compliant balloon catheter containing multiple impedance electrodes and a pressure transducer is positioned across the esophagogastric junction (EGJ). Stepwise or sustained balloon distension (usually 40 - 70 mL) generates luminal deformation that allows simultaneous measurement of cross-sectional area and intraballoon pressure along the esophageal axis. The data are represented as spatiotemporal maps of diameter, providing real-time visualization of EGJ opening and esophageal body contractile activity.12
This approach offers several physiologic advantages. Luminal distension activates intrinsic enteric reflexes, independent of the central mechanisms of swallowing, allowing for the assessment of neuromyogenic integrity even in patients unable to generate reliable swallows. In addition, distension-induced contractions follow the intrinsic latency gradient of the esophagus, revealing clinically relevant alterations in coordination, propagation, and contractile vigor that may not be apparent in HRM.9 - 11
Assessment of esophagogastric junction opening dynamics
A key strength of FLIP planimetry is its ability to quantify the opening dynamics of the EGJ. Traditional manometric metrics, such as integrated relaxation pressure (IRP), infer EGJ relaxation based on intraluminal pressure during swallowing. In contrast, FLIP directly measures the mechanical opening of the EGJ under distension, simultaneously integrating the effects of lower esophageal sphincter tone, crural diaphragm function, and esophageal wall compliance.
EGJ opening is characterized using two principal parameters: the EGJ distensibility index (EGJ-DI), calculated as the minimum cross-sectional area divided by intrabag pressure, and the maximum EGJ diameter reached during distension. These metrics have been validated against timed barium esophagram and in relation to clinical outcomes, demonstrating strong associations with clinically relevant obstruction.9, 11
Based on established thresholds, EGJ opening can be categorized as normal, borderline, or reduced. Normal opening is defined by an EGJ-DI ≥ 2.0 mm²/mmHg with a maximum diameter ≥ 16 mm, while reduced opening is defined by an EGJ-DI < 2.0 mm²/mmHg with a maximum diameter < 12 mm. Borderline opening included intermediate values that do not meet criteria for either category.12
It is essential to interpret EGJ opening in the context of esophageal contractile response. A mechanically adequate EGJ may nevertheless result in functional obstruction of bolus transit in patients with absent or severely impaired peristalsis, highlighting the need for integrated and contextualized interpretation rather than reliance on isolated metrics.
Contractile response patterns in FLIP planimetry
FLIP planimetry allows the esophageal contractile response to distension to be classified into reproducible patterns that correlate with clinically relevant motility phenotypes. The normal contractile response is defined by the presence of repetitive anterograde contractions (RACs) that meet the “rule of 6”: at least six consecutive anterograde contractions extending ≥ 6 cm in axial length, at a frequency of 6 ± 3 contractions per minute.10, 12
The borderline contractile response is characterized by distinguishable anterograde contractions that do not fully meet RAC criteria, while the impaired or disorganized response reflects absent or irregular contractile activity without organized propagation. The absent contractile response denotes complete lack of esophageal body contraction during distension.
A distinct category, known as the spastic-reactive contractile response, includes sustained occluding contractions, sustained contraction of the lower esophageal sphincter, or repetitive retrograde contractions. This pattern is strongly associated with spastic motility disorders, including type III achalasia, distal esophageal spasm, and hypercontractile esophagus (jackhammer).12
These FLIP-based phenotypes show substantial concordance with Chicago Classification v4.0 diagnoses, while providing complementary information, particularly in cases of equivocal HRM findings or incomplete manometric studies.
Integrating FLIP planimetry with endoscopic risk stratification
Role of Clinical Achalasia Risk Score (CARS)
While FLIP planimetry provides robust physiologic data, its diagnostic power is enhanced when interpreted in conjunction with structured endoscopic risk stratification. The Clinical Achalasia Risk Score (CARS) was developed to address the persistent diagnostic delay associated with achalasia by quantifying endoscopic features linked to esophageal outflow obstruction.9
The CARS incorporates four domains: esophageal contents, luminal anatomy, upper esophageal sphincter resistance, and stasis-related mucosal changes. Scores range from 0 to 8, with higher values indicating a greater likelihood of achalasia. Validation studies have demonstrated that a CARS score > 4 confers a greater than 95% probability of achalasia, while scores of 0-1 are associated with a very low probability of the disease.4
Probability-based diagnosis using CARS and FLIP
The integration of CARS and FLIP planimetry allows a probability-based diagnostic framework that transcends binary classification. Patients with high CARS scores and reduced EGJ opening on FLIP have an extremely high likelihood of achalasia and can proceed directly to definitive treatment without the need for confirmatory manometry. This approach is particularly relevant for patients with classic patterns of non-spastic obstruction, where diagnostic certainty is high. However, it should be noted that this approach should be reserved for scenarios with very high pretest probability, clinical-endoscopic-physiological concordance, and the availability of experienced personnel, given that high-resolution manometry remains the gold standard for formal motility classification and therapeutic definition in intermediate or discordant phenotypes.
Conversely, patients with low CARS scores and normal EGJ opening with preserved or borderline contractile response are unlikely to have a major motility disorder. In these cases, further evaluation should focus on GERD or gut-brain interaction disorders, rather than additional motility studies.
Intermediate scenarios -such as borderline EGJ opening, spastic-reactive contractile patterns, or moderate CARS scores- identify patients who benefit most from HRM or timed barium esophagram. It should be noted that studies suggest that only 30% to 50% of patients in this intermediate group ultimately meet criteria for a major motility disorder, underscoring the value of targeted indication for these studies rather than their routine use.5
In the proposed paradigm, centered on index endoscopy with FLIP planimetry, esophagram should be reserved for equivocal cases of suspected obstruction, particularly when diagnostic uncertainty persists after FLIP or when there is discordance between FLIP and high-resolution manometry. In patients with overt achalasia and high CARS scores, radiological evaluation rarely changes management. However, barium esophageal transit -particularly when performed with the timed protocol and tablet- continues to be clinically useful in evaluating bolus transit, the functional impact of obstruction, and the correlation with symptoms, especially in cases of intermittent dysphagia or when complementary dynamic assessment is required.Overall, this integrated approach would substantially reduce the need for catheter studies while maintaining high diagnostic accuracy.
Wireless pH monitoring during index endoscopy
Rationale and technical considerations
Wireless pH monitoring was developed to overcome the tolerance limitations associated with transnasal catheter reflux studies. The capsule is endoscopically affixed to the distal esophagus, typically 6 cm above the squamocolumnar junction, and allows for the recording of esophageal acid exposure over 48 to 96 hours. This extended monitoring period improves diagnostic performance by capturing the daily variability in reflux patterns.14
A particularly important point is that wireless pH monitoring can be performed during the same endoscopic session in which FLIP planimetry is conducted, eliminating the need for additional procedures. This is particularly advantageous in patients evaluated withoutf proton pump inhibitors, as recommended by contemporary guidelines when GERD diagnosis is uncertain.6
Role in the phenotyping of GERD and functional disorders
Objective assessment of reflux is essential for distinguishing pathologic GERD from reflux hypersensitivity and functional heartburn. Symptomatic response to PPIs alone is insufficient to establish diagnosis, given that the placebo response and non-acid mechanisms contribute substantially to the perception of symptoms.
Wireless pH monitoring allows accurate quantification of acid exposure time (AET) and facilitates correlation with patient-reported symptomatic events. According to the Lyon Consensus 2.0, abnormal AET (> 6%) is conclusive evidence of GERD, while a normal AET (< 4%) allows acid-mediated disease to be reliably excluded.14 Intermediate values require contextual interpretation, supported by adjunctive metrics or clinical correlation.
When integrated with FLIP planimetry and endoscopic assessment, wireless pH monitoring enables comprehensive phenotyping of patients with esophageal symptoms. Those with normal EGJ opening, preserved motility, and normal acid exposure are more accurately classified within the spectrum of gut-brain interaction disorders, guiding management toward neuromodulators, behavioral therapy, and reassurance strategies, rather than intensifying acid suppression.
In this context, a recent study conducted in Latin America showed that prolonged wireless pH monitoring allows for the reclassification of more than 70% of patients with borderline acid exposure in 24 - hour pH-impedance testing, overcoming the diagnostic “gray area” associated with catheter methods, which continue to be the most widely used in the region due to cost and access limitations.17
Clinical impact of an integrated strategy focused on index endoscopy
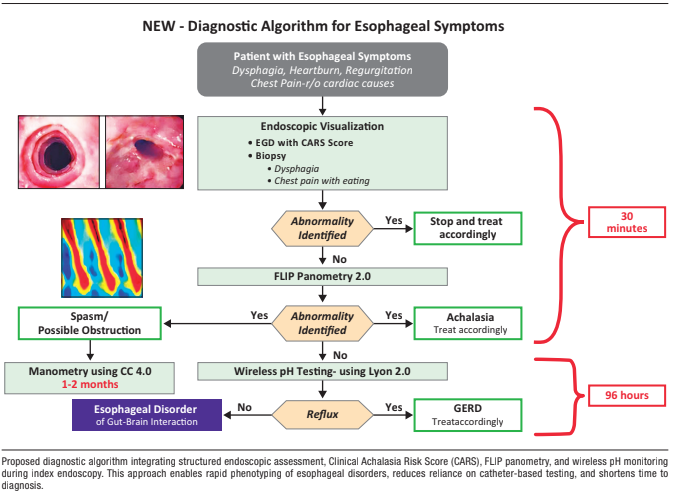
By incorporating FLIP planimetry, CARS scoring, and wireless pH monitoring into the index endoscopy, clinicians can establish a definitive diagnosis in most patients who would otherwise have an inconclusive evaluation. In the remaining patients, the physiological data obtained during endoscopy allow for targeted selection of confirmatory tests. Recent series from experienced centers have shown that this approach reduced the indication for manometry in a substantial proportion of patients and shortened the diagnostic time, although the magnitude of these benefits may vary depending on the local healthcare system (Figure 1). While available data suggest clinical and efficiency benefits, the magnitude of the impact may vary depending on local access to FLIP, wireless pH monitoring, and experience in physiological interpretation.
This strategy aligns with emerging cost-effectiveness data demonstrating that early physiological assessment is clinically superior and economically advantageous compared to empirical treatment and delayed diagnostics.15, 16 Furthermore, performing these assessments under sedation significantly improves the patient experience and study completion rates.
Pragmatic diagnostic algorithm centered on index endoscopy
Figure 2 presents a proposed diagnostic algorithm that repositions the initial endoscopy as the central diagnostic procedure for patients presenting with esophageal symptoms. This approach is based on the recognition that upper gastrointestinal endoscopy is inevitable in most cases, whether due to the presence of alarm symptoms, refractoriness to empirical treatment, or persistent diagnostic uncertainty, and takes advantage of this opportunity to incorporate a comprehensive physiological evaluation.
Figure 1. Diagnostic algorithm focused on index endoscopy
Figure 2. Clinical application of combined CARS and FLIP planimetry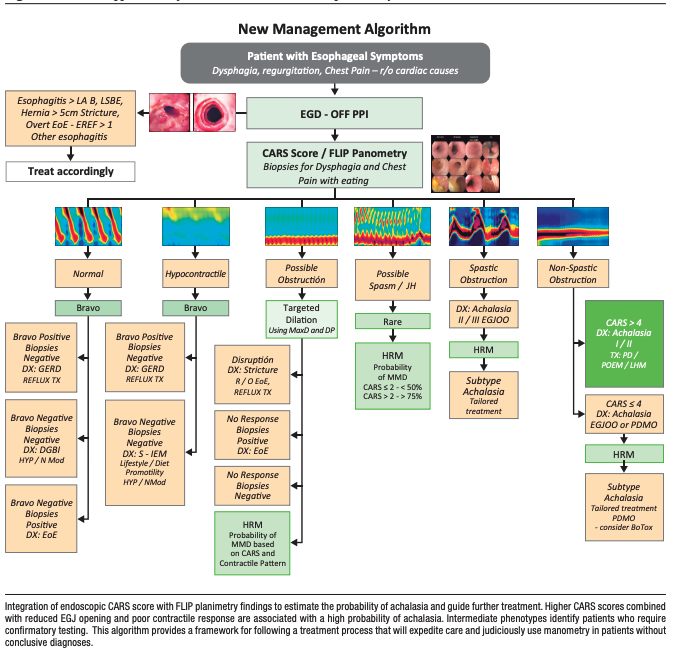
Patients presenting with dysphagia, regurgitation, food-related chest pain, or refractory heartburn undergo a high-quality upper gastrointestinal endoscopy, ideally without PPIs when feasible. The endoscopic evaluation includes systematic assessment of mucosal disease (Los Angeles classification and EREFS score), exclusion of mechanical obstruction, identification of anatomical abnormalities-such as hiatal hernia using the Hill classification-and evaluation of findings suggestive of achalasia using the Clinical Achalasia Risk Score (CARS).
When an obvious etiology is identified, such as grade B or higher esophagitis according to the Los Angeles classification, histologically confirmed eosinophilic esophagitis, fixed stenosis, malignancy, or large hiatal hernia,- management continues according to the established diagnosis, without the need for additional physiological tests. However, in the absence of a clear etiology, FLIP planimetry is performed during the same endoscopic session.
FLIP planimetry stratifies patients according to EGJ opening dynamics and contractile response. Patients with reduced EGJ opening and impaired or absent contractile response, particularly in the context of an elevated CARS score, have a high probability of presenting with achalasia and could proceed directly to definitive therapy without the need for confirmatory manometry. In contrast, patients with spastic-reactive contractile patterns could be referred for high-resolution manometry to define the subtype and guide a personalized therapeutic strategy.
Patients with normal EGJ opening and preserved or borderline contractile response have a low probability of having a major motility disorder. In these cases, wireless pH monitoring performed during index endoscopy allows for accurate phenotyping of GERD. Pathological acid exposure supports the diagnosis of GERD and guides treatment with acid suppression or anti-reflux interventions, while normal acid exposure favors the diagnosis of a gut–brain interaction disorder and directs management toward neuromodulation, behavioral interventions, and reassurance strategies.
This algorithm allows a definitive diagnosis to be established within a few days for most patients, minimizing diagnostic delays and reducing reliance on poorly tolerated catheter-based testing.
By consolidating diagnostic testing into a single instance, the proposed algorithm reduces procedural redundancy, shortens time to treatment, and improves patient satisfaction. These benefits are especially relevant for healthcare systems facing increasing demand, limited access to subspecialty testing, and pressure to reduce low-value clinical practices.
Figure 3. Clinical application based on combined CARS and FLIP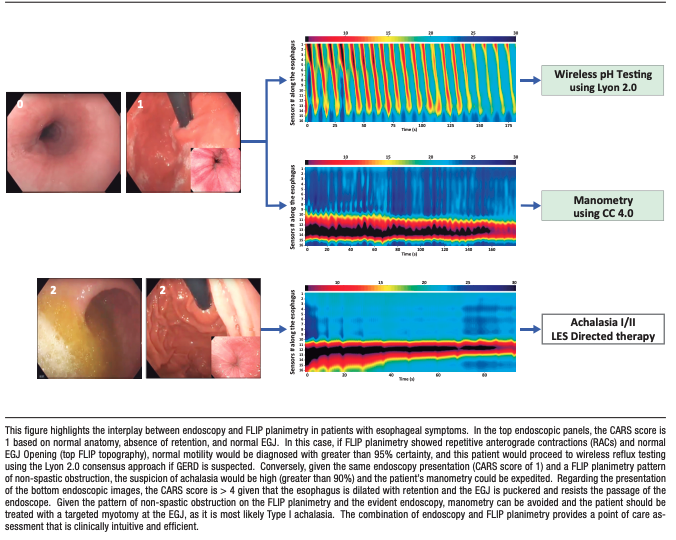
Figure 4.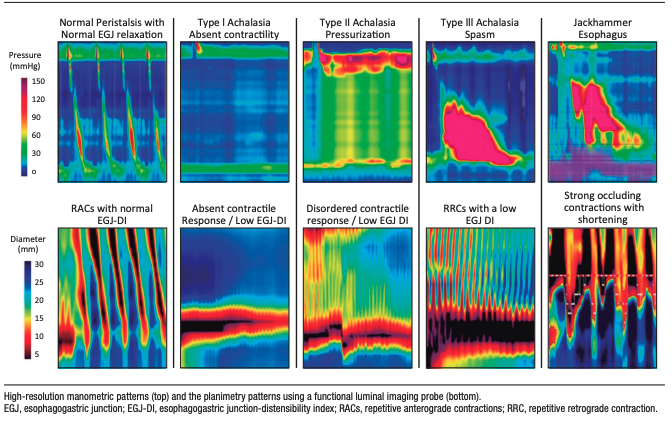
Future Directions
The integration of FLIP planimetry and structured endoscopic assessment represents only the first step toward precision esophagology. Current research aims to improve the physiological characterization of the esophagus by incorporating quantitative measurements of esophageal work and contractile power, which reflect how much energy the organ generates to propel the bolus, thus overcoming assessment based solely on pressure or diameter.
Artificial intelligence and machine learning offer promising avenues to enhance diagnostic accuracy and reproducibility. Automated interpretation of endoscopic findings, CARS scoring, and FLIP planimetry patterns could provide real-time decision support during endoscopy, reducing operator dependence and facilitating broader adoption of these diagnostic resources. Initial work using simplified mathematical models and computational simulations of the esophagus suggests that combining physiological data with computational modeling could enable greater treatment personalization, especially in complex motility disorders.
Future prospective studies are needed to validate outcome-based thresholds for FLIP planimetry, assess long-term clinical outcomes of algorithms focused on index endoscopy, and define optimal therapeutic options for intermediate phenotypes. As these data emerge, the role of endoscopy in esophageal disease is likely to evolve from a purely diagnostic tool to a platform for integrated physiological assessment and therapeutic decision-making.
Intellectual property. The authors declare that the data and figures presented in the manuscript are original and were carried out at their belonging institutions.
Funding. The authors declare that there were no external sources of funding.
Conflict of interest. The authors declare that they have no conflicts of interest in relation to this article.
Copyright

© 2026 Acta Gastroenterológica latinoamericana. This is an open-access article released under the terms of the Creative Commons Attribution (CC BY-NC-SA 4.0) license, which allows non-commercial use, distribution, and reproduction, provided the original author and source are acknowledged.
Cite this article as: Olmos J I y Pandolfino J E. Redefining the Initial Endoscopy Evaluation of Esophageal Symptoms: Toward a Precision-Based Diagnostic Paradigm. Acta Gastroenterol Latinoam. 2026;56(1):29-40. https://doi.org/10.52787/agl.v56i1.601
Correspondence: Juan Ignacio Olmos
Email: juan.ignacio.olmos@gmail.com
Acta Gastroenterol Latinoam 2026;56(1):29-40