Santiago Decotto ID· Rodolfo Pizarro ID
Department of Cardiology, Hospital Italiano de Buenos Aires.
Ciudad Autónoma de Buenos
Aires. Argentina.
Acta Gastroenterol Latinoam 2026;56(1):12-16
Received: 15/03/2026 / Accepted: 26/03/2026 / Published online: 31/03/2026 / https://doi.org/0.52787/agl.v56i1.619
Artificial intelligence (AI) has experienced exponential growth in recent years, driven by the development of machine learning algorithms, increased processing capacity, and the growing availability of large clinical databases.1-2 Its use has expanded into multiple areas of everyday clinical practice.3-4
Initially associated with image analysis or the automation of diagnostic processes, AI has broadened its scope and is now also involved in generating predictive models, risk stratification, and optimizing clinical decision. In parallel, scientific research has undergone a similar transformation: often, the magnitude and complexity of available data exceed the capabilities of traditional statistical approaches. In this context, AI emerges as a particularly useful tool for addressing this complexity. Accordingly, recent bibliometric analyses have documented a sustained increase in the number of publications and citations related to AI applied to healthcare in recent decades.1-5
AI is not limited to the analysis of large datasets, but can also support different stages of the research process, from hypothesis generation and methodological design to results analysis and even scientific writing.6
However, its incorporation is not without challenges. AI should be understood as a complementary resource that enhances scientific work, but does not replace clinical judgment or methodological rigor. Its responsible use requires understanding both its strengths and its limitations.7-8
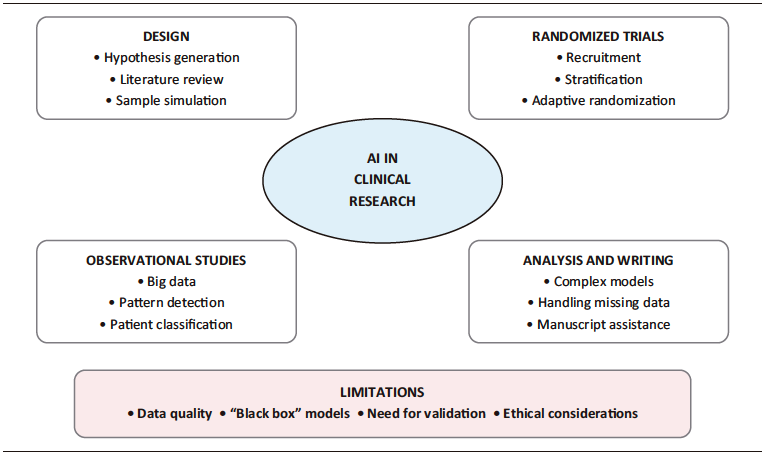
This brief review analyzes the main applications of AI in the design, execution, and analysis of clinical studies, distinguishing its contribution according to the type of methodological design and highlighting the considerations that must be taken into account for its appropriate implementation in research.
AI in the Research Study Design Phase
AI can play a relevant role from the earliest stages of the research process, even before patient recruitment or data collection begins. In the design phase, these tools can help optimize hypothesis formulation, methodological planning, and protocol structuring.
First, the automated analysis of large clinical databases allows for the identification of patterns, preliminary associations, or certain trends that can lead to new research questions. Unlike traditional approaches, machine learning–based methods can explore complex or non-linear relationships between variables, facilitating the generation of new data-driven hypotheses.9
Likewise, AI can assist in the review of the scientific literature, enabling broader and more structured searches, identifying relevant articles, detecting knowledge gaps, and synthesizing information in a preliminary way.10 While these tools do not replace the researcher’s critical appraisal, they can improve efficiency and reduce the time required for the initial analysis of a given topic.
Methodologically, AI can also be used to perform simulations that help estimate sample sizes or predict event rates under different assumptions. These simulations are particularly useful in studies with infrequent outcomes or in heterogeneous populations, where traditional planning involves greater uncertainties.11
Finally, in studies that use electronic health records or institutional registries, AI algorithms can assist in the operational definition of variables, the standardization of criteria, and the automated identification of potential participants who meet the predefined inclusion and exclusion criteria, potentially reducing errors resulting from manual case selection.12
AI in Randomized Clinical Trials
In randomized clinical trials, AI can be used at multiple stages of the study. As mentioned previously, algorithms applied to electronic health records allow for the automated identification of potentially eligible patients based on predefined criteria, optimizing time and reducing recruitment bias.12 Predictive models can also contribute to better baseline risk stratification, promoting a more balanced allocation of participants.
In more complex designs, AI can be integrated into adaptive randomization schemes, an approach in which the probability of assignment to each arm is dynamically adjusted throughout the study based on accumulated interim results. This type of design aims to improve trial efficiency and optimize the use of information generated during its development.13-14 Furthermore, automated tools already exist that can facilitate the monitoring of adverse events and the early detection of protocol deviations.
While these applications do not modify the fundamental principles of clinical trials, they can improve operational efficiency and the quality of data control.
AI in Observational Studies and Big Data Analysis
In observational studies, both prospective and retrospective, AI offers useful tools for analyzing large volumes of information.
In prospective cohorts, it can be used during follow-up to update risk estimates, incorporate new variables over time, and facilitate the automated identification of clinical events.
In retrospective studies and large database analyses, areas where AI has likely had the greatest impact, its usefulness lies in its ability to organize and analyze large volumes of already available information. In recent years, retrospective studies based on national or international databases have become increasingly common, particularly in countries with fully digitized information systems. Many of these studies are published in high-impact medical journals and provide evidence that, in some cases, is considered for the development or updating of clinical practice guidelines.
In this context, AI-based methods allow for the review of thousands of records in a short time, the identification of associations between multiple variables, and the detection of patterns that would be difficult to recognize manually. They can also help classify patients into groups with similar characteristics and extract relevant information from clinical records or reports.15 These tools expand the possibilities for analysis and can generate new research hypotheses. However, the results obtained using these methods should be interpreted with caution and validated in other populations to avoid drawing erroneous conclusions or identifying non-reproducible associations.
AI in Statistical Analysis and Scientific Writing
In addition to its role in study design and data collection, AI can play a significant role in the statistical analysis stage, especially when working with many variables or diverse information. These methods allow for the simultaneous analysis of multiple factors and the exploration of their interrelationships, facilitating the development of prognostic models and the integration of clinical data, laboratory results, imaging, or electronic records within a single analysis.
One particularly useful application is the handling of missing data, a common issue in real-world practice. Instead of simply excluding cases or replacing values, algorithms can estimate missing information by considering the full set of available variables. This can help reduce bias and improve the robustness of the results.16
Additionally, clustering techniques allow for the identification of subgroups of patients with similar characteristics within a given cohort. This can be useful for recognizing different clinical profiles, better understanding the heterogeneity of certain diseases, and generating new research hypotheses.
However, the use of these tools requires caution. A model should not be considered valid simply because it performs well on the data used to develop it. Its performance must be evaluated in independent populations to ensure that results are consistent and applicable in other settings. It is also important to avoid overfitting to the original dataset, as this may limit its practical utility.
On the other hand, various AI-based tools applied to scientific writing can assist in organizing the manuscript, improving textual clarity, and facilitating grammatical correction or translation into other languages.17
Limitations of AI in Research
Although AI offers multiple advantages, it also presents limitations that must be considered. First, the results depend heavily on the quality of the data used. If the information is incomplete, unrepresentative, or contains errors, models may generate unreliable conclusions or findings that are difficult to apply in other contexts.
Additionally, some algorithms function as “black boxes.” This means that although the model may generate a highly accurate prediction or recommendation, the internal process by which it reaches that conclusion is not always transparent or easily understandable, and may even be illogical. This failure to interpret the analysis process can hinder the understanding of findings, limit confidence in the results, and represent a barrier to their application in clinical practice.18
Beyond these technical limitations, there are relevant ethical considerations. The use of large clinical databases requires ensuring the protection of privacy and data confidentiality of information, as well as compliance with current regulatory frameworks. The Central Figure illustrates the current role of AI in clinical research, integrating its main methodological applications and associated challenges.
Artificial Intelligence Tools Applied to the Clinical Research Process
In recent years, various AI-based tools have emerged that can assist researchers at different stages of the scientific process, from literature search to the visual communication of results. In the initial phase, platforms such as OpenEvidence and SciSpace enable semantic searches of the scientific literature, summarization of articles, and efficient extraction of relevant information, facilitating the identification of knowledge gaps and the formulation of new research questions.
During methodological design and study planning, language models such as ChatGPT and Claude can assist in structuring hypotheses, defining variables, and reviewing the coherence of the protocol. In the data analysis stage, these tools can also be used to support script generation in statistical environments such as R or Python, the exploration of complex datasets, and the preliminary interpretation of results.
Similarly, in scientific writing, they can help improve text clarity, organize the manuscript, and facilitate translation into other languages. Finally, for the visual communication of findings, platforms such as BioRender, Canva, Gamma, or SciSpace allow the creation of diagrams, scientific figures, and visual abstracts that enhance the dissemination of results.
Although these tools can increase the efficiency of the research process, their use should be considered complementary to the researcher’s clinical judgment and methodological rigor (Table 1).
Central Figure. Artificial Intelligence in Clinical Research
Table 1. Artificial intelligence tools applied to different stages of the clinical
research process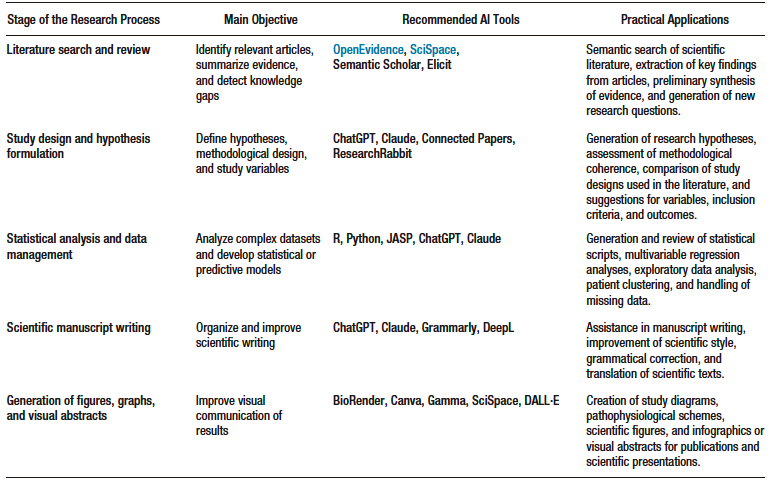
AI has been progressively incorporated into clinical research and offers useful tools to improve efficiency, the analysis of complex data, and scientific production. Its ability to process large volumes of information expands research possibilities across different methodological designs.
However, its true value lies in its responsible and complementary use. When integrated with scientific judgment and academic oversight, it can help strengthen the quality of evidence without replacing the fundamental principles of clinical research.
Intellectual property. The authors declare that the data, figures and tables in this article are original and were carried out at their institutions.
Funding. The authors declare that there were no external sources of funding.
Conflict of interest. The authors declare that they have no conflicts of interest in relation to this article.
Copyright

© 2026 Acta Gastroenterológica latinoamericana. This is an open-access article released under the terms of the Creative Commons Attribution (CC BY-NC-SA 4.0) license, which allows non-commercial use, distribution, and reproduction, provided the original author and source are acknowledged.
Cite this article as: Decotto S y Pizarro R. Role of Artificial Intelligence in Clinical Research: Methodological Applications and Current Challenges. 2026;56(1):12-16. https://doi.org/0.52787/agl.v56i1.619
Correspondence: Santiago Decotto
Email: santiago.decotto@hospitalitaliano.org.ar
Acta Gastroenterol Latinoam 2026;56(1):12-16