Pre- and Post-Processing Evaluations of Manual and Automated Endoscopes: A Microbiological Isolation
DOI:
https://doi.org/10.52787/agl.v54i3.423Keywords:
Endoscopes, reprocessing, contaminationAbstract
Introduction. An endoscope is a flexible tube equipped with a camera and a light used for the diagnosis and treatment of digestive pathologies. Although healthcare-associated infections are uncommon, they represent a significant concern. Proper endoscope reprocessing is essential to prevent infections, requiring adherence to established guidelines for cleaning, disinfection, and storage.
Objetive. To compare the effectiveness of manual and automated reprocessing of video gastroscopes and colonoscopes through microbiological monitoring of the following microorganisms: ESBL Escherichia coli, Shigella spp., Salmonella spp., Pseudomonas aeruginosa, Klebsiella pneumoniae carbapenemase (KPC)-producing, and vancomycin-resistant Enterococcus spp.
Materials and Methods. A prospective, observational, descriptive study was conducted, in which 207 reprocessing procedures of video gastroscopes and colonoscopes were selected for evaluation. Of these, 103 were manual and 104 were automated. The procedures were evaluated in both the pre- and post-processing stages. Cultures were performed to isolate ESBL Escherichia coli, Shigella spp., Salmonella spp., Pseudomonas aeruginosa, Klebsiella pneumoniae carbapenemase (KPC)-producing, and vancomycin-resistant Enterococcus spp.
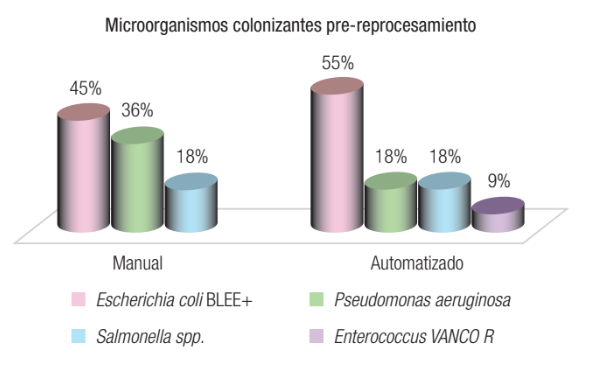
Results. In the pre-reprocessing samples, ESBL Escherichia coli was isolated in 45% of cases, compared to 55% for manual and automated reprocessing, respectively. Similarly, Pseudomonas aeruginosa was isolated in 36% of cases for manual reprocessing and 18% for automated reprocessing. Salmonella spp. was isolated in 18% of cases for both manual and automated reprocessing. Finally, vancomycin-resistant Enterococcus spp. was isolated in 9% of automated reprocessing samples. The results of the manual post-processing demonstrated that 1% of the samples exhibited counts of 1,000–99,999 colony-forming units (CFU)/ml, while 6% exhibited counts exceeding 100,000 CFU/ml. The automated method demonstrated a 7% prevalence of samples with over 100,000 colony-forming units (CFU) per milliliter. A comparison of the two modalities revealed that pathogens were not identified in seven of the 103 manual samples and six of the 104 automated samples. The only growth observed in the automated samples was that of coagulase-negative Staphylococcus, which is a potential contaminant. However, one automated sample did yield Salmonella spp.
Conclusion. The results obtained were comparable between the two methodologies. The automated reprocessing revealed the presence of an enteropathogen, which will allow for a reassessment of the reprocessing steps in order to enhance the efficacy of the procedures.
References
-1. Blázquez-Garrido RM, Cuchí-Burgos E, Martín-Salas C, Ruiz-Garbajosa P. Métodos microbiológicos para la monitorización de la limpieza, desinfección y esterilización de dispositivos médicos. Enferm Infecc Microbiol Clin [Internet]. 2018;36(10):657-61. Disponible en: http://dx.doi.org/10.1016/j.eimc.2017.09.012
-2. Juan H. Programa Nacional de Vigilancia de Infecciones Hospitalarias de Argentina (VIHDA). Áreas Críticas y Cirugía Programa Nacional. 2019;2020.
-3. Kovaleva J, Peters FT, Van Der Mei HC, Degener JE. Transmission of infection by flexible gastrointestinal endoscopy and bronchoscopy. Clinical Microbiology Reviews. 2013;26(2):231-54.
-4. Nelson DB. Infectious disease complications of Gl endoscopy: Part I, endogenous infections. Gastrointestinal Endoscopy. 2003;57(4):546-56.
-5. Lascano VM. Control de calidad microbiológico en la desinfección de colonoscopios, gastroscopios y broncoscopios. Trabajo Integrador Final (Especialista en Esterilización). Facultad de Ciencias Químicas. 2015.
-6. Robles C, Turín C, Villar A, Huerta-Mercado J, Samalvides F. Evaluación microbiológica de la desinfección de alto nivel de los endoscopios flexibles en un hospital general. Revista de Gastroenterología del Perú. 2014;34(2): 115-9:
-7. Rutala WA, Weber DJ. Guideline for Disinfection and Sterilization in Healthcare Facilities. North Carolina: CDC. 2008;158.
-8. Speer T. Actualización de la desinfección de endoscopios: guía para un reprocesamiento sensible a los recursos. WGO/WEO Global Guideline Endoscope Disinfection. World Gastroenterology Organisation. 2019.
-9. Diana Weinstock, Nadia Poczter, Lucía Daciuk y Analía Martínez. Consenso entre las siguientes Sociedades Científicas: Endoscopistas Digestivos de Buenos Aires-Sociedad Argentina de Infectología - Club Argentino de Esterilización. Guía de reprocesamiento de endoscopios y material accesorios [Internet]. 2014. Disponible en: http://www.endiba.org.ar/site/index.php/recursos/guias.
-10. Beilenhoff U, Neumann CS, Rey JF, Biering H, Blum R, Schmidt V, & null and the ESGE Guidelines Committee. ESGE-ESGENA guideline for quality assurance in reprocessing: microbiological surveillance testing in endoscopy. Endoscopy. 2007;39(02):175-81.
-11. Graziano KU, Pereira MEA, Koda E. Propuesta metodológica para la validación de la eficacia de la desinfección de un reprocesador automatizado de endoscopios flexibles. Revista Latino-Americana de Enfermagem. 2016.
-12. Sosa-Hernández Ó, Campos-Jiménez R. Relevancia del reprocesamiento de los endoscopios en el control de infecciones. Endoscopia [Internet]. 2019;31(4). Disponible en: http://dx.doi.org/10.24875/end.19000023.
-13. Shin SP, Kim WH. Recent update on microbiological monitoring of gastrointestinal endoscopes after High-Level disinfection. Clin Endosc [Internet]. 2015;48(5):369-73. Disponible en: http://dx.doi.org/10.5946/ce.2015.48.5.369.
-14. Manual sobre el método de endoscopía. Instrucciones de utilización asociadas con el sistema [Internet]. Disponible en: https:www.olympusoste.eu/media/contact_and_support/download/system_guides/W7052_803.pdf.
-15. Petersen BT, Chennat J, Cohen J, Cotton PB, Greenwald DA, Kowalski TE, et al. Multisociety guideline on reprocessing flexible GI endoscopes: 2011. Infect Control Hosp Epidemiol [Internet]. 2011;32(6):527-37. Disponible en: http://dx.doi.org/10.1086/660676.
-16. Spaulding EH, Groschel DH. Hospital disinfectants and antiseptics. Manual of clinical microbiology. Washington DC: American Society for Microbiology. 1974. Pp. 852-7.
-17. Argaraña Gerico A, Enriqueta H-S. Recomendaciones AEEED Limpieza y desinfección en endoscopía gastrointestinal. www.aeeed.com; 2013.
-18. Bradley CR, Babb JR. Endoscope decontamination: automated vs. manual. J Hosp Infect [Internet]. 1995;30Suppl:537-42. Disponible en: http://dx.doi.org/10.1016/0195-6701(95)90060-8 .
-19. Pérez DN, González IB, Pastor VP, Mateu PN, Bellver VO, Felis TS. y Viudez L A, editor. Comparación de la eficacia en la limpieza y desinfección de videoendoscopios: mecanizada frente a manual. 2000 23(9): 412-5.
-20. Alfa MJ, Olson N, DeGagne P. Automated washing with the Reliance Endoscope Processing System and its equivalence to optimal manual cleaning. Am J Infect Control [Internet]. 2006;34(9):561-70. Disponible en: http://dx.doi.org/10.1016/j.ajic.2006.01.010.
-21. Kaczmarek RG, Moore RM Jr, McCrohan J, Goldmann DA, Reynolds C, Caquelin C, et al. Multi-state investigation of the actual disinfection/sterilization of endoscopes in health care facilities. Am J Med [Internet]. 1992;92(3):257-61. Disponible en: http://dx.doi.org/10.1016/0002-9343(92)90074-l.
-22. Acosta-Gnass SI. Manual de esterilización para centros de salud. Pan American Health Org. 2008.
Bader L, Blumenstock G, Birkner B, Leiss O, Heesemann J, Riemann JF, et al. HYGEA (Hygiene in gastroenterology-endoscope reprocessing): Study on quality of reprocessing flexible endoscopes in hospitals and in the practice setting. Zeitschrift fur Gastroenterologie. 2002;40(3):157-70.
Published
How to Cite
Issue
Section
License
Copyright (c) 2024 Romina Medeot, Javier Mena, Pablo Rodríguez, Pablo Herrera Najum, Luis López, María Soledad Muñoz

This work is licensed under a Creative Commons Attribution-NonCommercial-ShareAlike 4.0 International License.












