Advances in the Treatment of Refractory Pediatric Constipation
DOI:
https://doi.org/10.52787/agl.v56i1.576Keywords:
Refractory functional constipation, pediatrics, fecal incontinence, laxatives, transanal irrigation, sacral and posterior tibial nerve stimulation, botulinum toxinAbstract
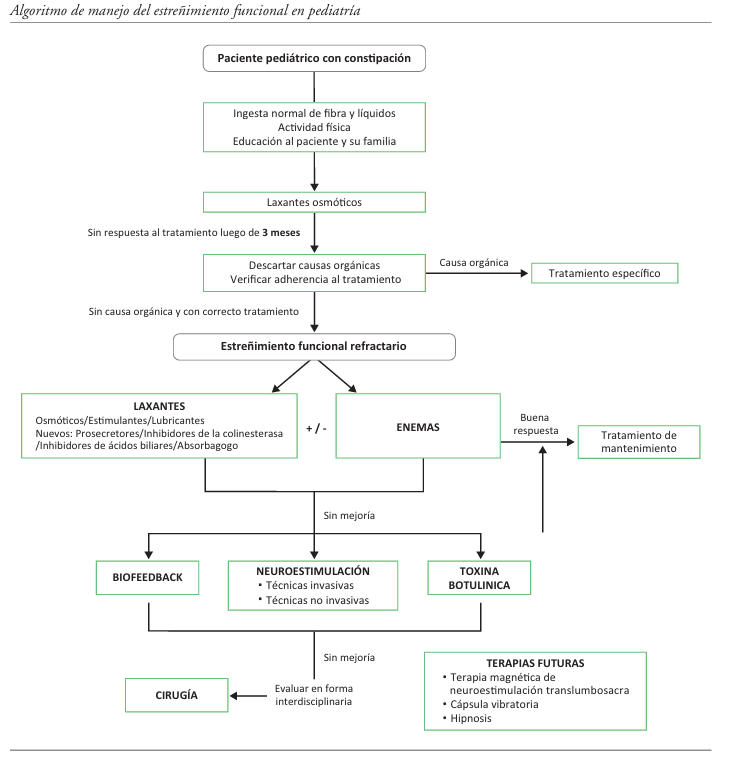
Refractory functional constipation is a persistent challenge in pediatrics. Beyond polyethylene glycol and traditional laxatives, new pharmacological and non-pharmacological therapies are emerging. Among the innovative medications, prosecretory agents like linaclotide (already approved for children over 6 years old), lubiprostone and plecanatide, which increase intestinal secretion, stand out. Other drugs, such as prucalopride (5HT-4 agonist) and pyridostigmine, aim to improve motility. Rectal and interventional therapies include transanal irrigation -which has shown good results-, antegrade enemas and botulinum toxin A injections, which are useful for specific dysfunctions. Biofeedback remains an effective tool. Neuromodulation is emerging as a promising option. Implanted sacral nerve stimulation and non-invasive techniques such as percutaneous/transcutaneous tibial nerve stimulation and transcutaneous sacral neuromodulation demonstrate improvements in symptoms and quality of life. There are therapies that have been little explored but show promise for the future, such as vibrating capsules, magnetic neurostimulation, and mind-body approaches like hypnosis. Specific pediatric research and an individualized treatment approach are crucial to optimize adherence and outcomes in these complex patients.
References
-1. Koppen IJN, Vriesman MH, Saps M, et al. Prevalence of functional defecation disorders in children: a systematic review and meta-analysis. J Pediatr. 2018;198:121-130.e6. DOI: 10.1016/j.jpeds.2018.02.029
-2. Rasquin A, Di Lorenzo C, Forbes D, et al. Childhood functional gastrointestinal disorders: child/adolescent. Gastroenterology. 2006;130(5):1527-1537. DOI: 10.1053/j.gastro.2005.08.063
-3. Koppen IJ, Nurko S, Saps M, et al. The pediatric Rome IV criteria: what’s new?. Expert Rev Gastroenterol Hepatol. 2017;11(3):193-201. DOI: 10.1080/17474124.2017.1282820
-4. Wolfson S, Saps M. Recent advances in treating constipation in children. Expert Rev Gastroenterol Hepatol. 2024;18(7):325-338. DOI: 10.1080/17474124.2024.2383636. PMID: 39034812
-5. Tabbers MM, DiLorenzo C, Berger MY, Faure C, Langendam MW, Nurko S, et al. Evaluation and treatment of functional constipation in infants and children: evidence-based recommendations from ESPGHAN and NASPGHAN. J Pediatr Gastroenterol Nutr. 2014;58(2):258-274. DOI: 10.1097/mpg.0000000000000266
-6. Bardisa-Ezcurra L, Ullman R, Gordon J. Diagnosis and management of idiopathic childhood constipation: summary of NICE guidance. BMJ. 2010;340:c2585. DOI: 10.1136/bmj.c2585
-7. van Ginkel R, Reitsma JB, Buller HA, et al. Childhood constipation: longitudinal follow-up beyond puberty. Gastroenterology. 2003;125(2):357-363. DOI: 10.1016/S0016-5085(03)00888-6
-8. Pijpers MA, Bongers ME, Benninga MA, Berger MY. Functional constipation in children: a systematic review on prognosis and predictive factors. J Pediatr Gastroenterol Nutr. 2010;50(3):256-268. DOI: 10.1097/MPG.0b013e3181afcdc3
-9. Gordon M, MacDonald JK, Parker CE, Akobeng AK, Thomas AG. Osmotic and stimulant laxatives for the management of childhood constipation. Cochrane Database Syst Rev. 2016;(8):CD009118. DOI: 10.1002/14651858.CD009118.pub3
-10. Bonilla S, Nurko S, Rodriguez L. Long-term use of Bisacodyl in pediatric functional constipation refractory to conventional therapy. J Pediatr Gastroenterol Nutr. 2020;71(3):288-291. DOI: 10.1097/MPG.0000000000002795
-11. Koppen IJ, Lammers LA, Benninga MA, et al. Management of functional constipation in children: therapy in practice. Paediatr Drugs. 2015;17(5):349-360. DOI: 10.1007/s40272-015-0142-4
-12. Chen JJ, Kitzia Colliard RN, Nurko S, et al. Melanosis Coli is not associated with colonic dysmotility nor severity of pediatric functional constipation. Dig Dis Sci. 2022;67(8):3922-3928. DOI: 10.1007/s10620-021-07191-z
-13. Urganci N, Akyildiz B, Polat TB. A comparative study: the efficacy of liquid paraffin and lactulose in management of chronic functional constipation. Pediatr Int. 2005;47(1):15-19.
-14. Clark JH, Russell GJ, Fitzgerald JF, et al. Serum beta-carotene, retinol, and alpha-tocopherol levels during mineral oil therapy for constipation. Am J Dis Child. 1987;141(11):1210-1212. DOI: 10.1001/archpedi.1987.04460110080028
-15. Bijvelds MJC, Bot AGM, Escher JC, et al. Activation of intestinal Cl- secretion by lubiprostone requires the cystic fibrosis transmembrane conductance regulator. Gastroenterology. 2009;137(3):976-985. DOI: 10.1053/j.gastro.2009.05.037
-16. Fukudo S, Hongo M, Kaneko H, et al. Lubiprostone increases spontaneous bowel movement frequency and quality of life in patients with chronic idiopathic constipation. Clin Gastroenterol Hepatol. 2015;13(2):294-301.e5. DOI: 10.1016/j.cgh.2014.08.026
-17. Hyman PE, Di Lorenzo C, Prestridge LL, et al. Lubiprostone for the treatment of functional constipation in children. J Pediatr Gastroenterol Nutr. 2014;58(3):283-291. DOI: 10.1097/MPG.0000000000000176
-18. Benninga MA, Hussain SZ, Sood MR, et al. Lubiprostone for pediatric functional constipation: randomized, controlled, double-blind study with long-term extension. Clin Gastroenterol Hepatol. 2022;20(3):602-610.e5. DOI: 10.1016/j.cgh.2021.04.005
-19. FDA approves first treatment for pediatric functional constipation. 2023 [cited 2024 Feb 18]. Available from: https://www.fda.gov/drugs/news-events-human-drugs/fda-approves-first-treatment-pediatric-functional-constipation
-20. Di Lorenzo C, Nurko S, Hyams JS, et al. Randomized controlled trial of linaclotide in children aged 6-17 years with functional constipation. J Pediatr Gastroenterol Nutr. 2024;78(5):1059-1068. DOI: 10.1002/jpn3.12184
-21. Barish CF, Griffin P. Safety and tolerability of plecanatide in patients with chronic idiopathic constipation: long-term evidence from an open-label study. Curr Med Res Opin. 2018;34(4):751-755. DOI: 10.1080/03007995.2018.1430024
-22. Kamuda JA, Mazzola N. Plecanatide (Trulance) for chronic idiopathic constipation and irritable bowel syndrome with constipation. Pharm Ther. 2018;43(4):207-232
-23. Shailubhai K, Comiskey S, Foss JA, et al. Plecanatide, an oral guanylate cyclase C agonist acting locally in the gastrointestinal tract, is safe and well-tolerated in single doses. Dig Dis Sci. 2013;58(9):2580-2586. DOI: 10.1007/s10620-013-2684-z
-24. Barish CF, Griffin P. Safety and tolerability of plecanatide in patients with chronic idiopathic constipation: long-term evidence from an open-label study. Curr Med Res Opin. 2018;34(4):751-755. DOI: 10.1080/03007995.2018.1430024
-25. Miner PB Jr, Koltun WD, Wiener GJ, De La Portilla M, Prieto B, Shailubhai K, et al. A randomized phase III clinical trial of plecanatide, a uroguanylin analog, in patients with chronic idiopathic constipation. Am J Gastroenterol. 2017;112(4):613-621. DOI: 10.1038/ajg.2016.611
-26. Sajid MS, Hebbar M, Baig MK, et al. Use of prucalopride for chronic constipation: a systematic review and meta-analysis of published randomized, controlled trials. J Neurogastroenterol Motil. 2016;22(3):412-422. DOI: 10.5056/jnm16004
-27. Kaufman MB. Pharmaceutical approval update. Pharm Ther. 2019;44(4):178-209
-28. Winter HS, Di Lorenzo C, Benninga MA, et al. Oral prucalopride in children with functional constipation. J Pediatr Gastroenterol Nutr. 2013;57(2):197-203. DOI: 10.1097/MPG.0b013e318292f9ea
-29. Mugie SM, Korczowski B, Bodi P, et al. Prucalopride is no more effective than placebo for children with functional constipation. Gastroenterology. 2014;147(6):1285-1295.e1. DOI: 10.1053/j.gastro.2014.09.005
-30. Koppen IJ, Lammers LA, Benninga MA, Tabbers MM. Management of functional constipation in children: therapy in practice. Paediatr Drugs. 2015;17(5):349-360. DOI: 10.1007/s40272-015-0142-4
-31. Gordon M, Grafton-Clarke C, Rajindrajith S, Benninga MA, Sinopoulou V, Akobeng AK. Treatments for intractable constipation in childhood. Cochrane Database Syst Rev. 2024;19(6):CD014580. DOI: 10.1002/14651858.CD014580.pub2. PMID: 38895907; PMCID: PMC11190639
-32. Manini ML, Camilleri M, Grothe R, et al. Application of pyridostigmine in pediatric gastrointestinal motility disorders: a case series. Paediatr Drugs. 2018;20(2):173-180. DOI: 10.1007/s40272-017-0277-6
-33. Soufi-Afshar I, Moghadamnia A, Bijani A, et al. Comparison of pyridostigmine and bisacodyl in the treatment of refractory chronic constipation. Caspian J Intern Med. 2016;7(1):19-24
-34. O'Dea CJ, Brookes JH, Wattchow DA. The efficacy of treatment of patients with severe constipation or recurrent pseudo-obstruction with pyridostigmine. Colorectal Dis. 2010;12(6):540-548. DOI: 10.1111/j.1463-1318.2009.01838.x. PMID: 19508545
-35. Manini ML, Camilleri M, Grothe R, Di Lorenzo C. Application of pyridostigmine in pediatric gastrointestinal motility disorders: a case series. Paediatr Drugs. 2018;20(2):173-180. DOI: 10.1007/s40272-017-0277-6. PMID: 29243034
-36. Nakajima A, Ishizaki S, Matsuda K, et al. Impact of elobixibat on serum and fecal bile acid levels and constipation symptoms in patients with chronic constipation. J Gastroenterol Hepatol. 2022;37(5):883-890. DOI: 10.1111/jgh.15800
-37. Khanna L, Camilleri M. Review article: elobixibat: a novel treatment for chronic constipation. Aliment Pharmacol Ther. 2021;53(2):234-242. DOI: 10.1111/apt.16143
-38. Wong BS, Camilleri M. Elobixibat for the treatment of constipation. Expert Opin Investig Drugs. 2013;22(2):277-284. DOI: 10.1517/13543784.2013.753056
-39. Spencer AG, Labonte ED, Rosenbaum DP, et al. Intestinal inhibition of the Na+/H+ exchanger 3 prevents cardiorenal damage in rats and inhibits Na+ uptake in humans. Sci Transl Med. 2014;6(227):227ra36. DOI: 10.1126/scitranslmed.3007790
-40. Chey WD, Lembo AJ, Rosenbaum DP. Efficacy of tenapanor in treating patients with irritable bowel syndrome with constipation: a 12-week, placebo-controlled phase 3 trial (T3MPO-1). Am J Gastroenterol. 2020;115(2):281-293. DOI: 10.14309/ajg.0000000000000516
-41. Nasher O, Hill RE, Peeraully R, et al. Peristeen® transanal irrigation system for paediatric faecal incontinence: a single centre experience. Int J Pediatr. 2014;2014:954315. DOI: 10.1155/2014/954315
-42. Patel S, Hopson P, Bornstein J, et al. Impact of transanal irrigation device in the management of children with fecal incontinence and constipation. J Pediatr Gastroenterol Nutr. 2020;71(3):292-297. DOI: 10.1097/MPG.0000000000002785
-43. Midrio P, Mosiello G, Ausili E, et al. Peristeen® transanal irrigation in paediatric patients with anorectal malformations and spinal cord lesions: a multicentre Italian study. Colorectal Dis. 2016;18(1):86-93. DOI: 10.1111/codi.13101
-44. Baaleman DF, Wegh CAM, Hoogveld MTA, et al. Transanal irrigation in children: treatment success, quality of life, adherence, patient experience, and independence. J Pediatr Gastroenterol Nutr. 2022;75(2):166-172. DOI: 10.1097/MPG.0000000000003515
-45. Koppen IJ, Kuizenga-Wessel S, Voogt HW, et al. Transanal irrigation in the treatment of children with intractable functional constipation. J Pediatr Gastroenterol Nutr. 2017;64(2):225-229. DOI: 10.1097/MPG.0000000000001236
-46. Ng J, Ford K, Dalton S, et al. Transanal irrigation for intractable faecal incontinence and constipation: outcomes, quality of life and predicting non-adopters. Pediatr Surg Int. 2015;31(8):729-734. DOI: 10.1007/s00383-015-3735-7
-47. Pacilli M, Pallot D, Andrews A, et al. Use of Peristeen® transanal colonic irrigation for bowel management in children: a single-center experience. J Pediatr Surg. 2014;49(2):269-272. DOI: 10.1016/j.jpedsurg.2013.11.036
-48. Christensen P, Krogh K, Buntzen S, et al. Long-term outcome and safety of transanal irrigation for constipation and fecal incontinence. Dis Colon Rectum. 2009;52(2):286-292. DOI: 10.1007/DCR.0b013e3181979341
-49. Tamvakeras P, Horrobin C, Chang J, et al. Long-term outcomes of transanal irrigation for bowel dysfunction. Cureus. 2023;15(7):e42507. DOI: 10.7759/cureus.42507
-50. Hamonet-Torny J, Bordes J, Daviet JC, et al. Long-term transanal irrigation’s continuation at home. Preliminary study. Ann Phys Rehabil Med. 2013;56(2):134-142. DOI: 10.1016/j.rehab.2013.01.005
-51. Baaleman DF, Vriesman MH, Lu PL, et al. Long-term outcomes of antegrade continence enemas to treat constipation and fecal incontinence in children. J Pediatr Gastroenterol Nutr. 2023;77(2):191-197. DOI: 10.1097/MPG.0000000000003833
-52. Koivusalo A, Pakarinen MP, Rintala RJ. Treatment of a leaking ACE conduit with deflux injections. Pediatr Surg Int. 2006;22(12):1003-1006. DOI: 10.1007/s00383-006-1792-7
-53. Sinha CK, Grewal A, Ward HC. Antegrade continence enema (ACE): current practice. Pediatr Surg Int. 2008;24(6):685-688. DOI: 10.1007/s00383-008-2130-z
-54. Siddiqui AA, Fishman SJ, Bauer SB, et al. Long-term follow-up of patients after antegrade continence enema procedure. J Pediatr Gastroenterol Nutr. 2011;52(5):574-580. DOI: 10.1097/MPG.0b013e3181ff6042
-55. Vernamonti JP, Hauck C, Santos EP, Wild LC, Ralls MW, Jarboe MD, Speck KE, Ehrlich PF. Do preoperative factors predict success of antegrade continence enemas in children?. J Pediatr Surg. 2023;58(1):52-55. DOI: 10.1016/j.jpedsurg.2022.09.029
-56. Langer JC, Birnbaum E. Preliminary experience with intrasphincteric botulinum toxin for persistent constipation after pull-through for Hirschsprung’s disease. J Pediatr Surg. 1997;32(7):1059-1061. DOI: 10.1016/S0022-3468(97)90399-7
-57. Keshtgar AS, Suliman A, Thakkar H, Selim I. Long-term outcomes of botulinum toxin injection into the external anal sphincters: an effective new treatment of chronic functional constipation in children. J Pediatr Surg. 2025;60(3):162049. DOI: 10.1016/j.jpedsurg.2024.162049
-58. Zar-Kessler C, Kuo B, Belkind-Gerson J. Botulinum toxin injection for childhood constipation is safe and can be effective regardless of anal sphincter dynamics. J Pediatr Surg. 2018;53(4):693-697. DOI: 10.1016/j.jpedsurg.2017.12.007
-59. Baaleman DF, Hallagan A, Halleran DR, et al. Anal botulinum toxin in children with Hirschsprung disease and functional constipation: a prospective cohort study. Eur J Pediatr Surg. 2023;33(3):241-248. DOI: 10.1055/s-0042-1751048
-60. Radwan AB, Gadallah MA, Shahawy MR, et al. Can botulinum toxin help in managing children with functional constipation and obstructed defecation?. J Pediatr Surg. 2021;56(4):750-753. DOI: 10.1016/j.jpedsurg.2020.06.044
-61. Walther M, Muller H, Weiss C, et al. A comparative observational study on intrasphincteric injections with botulinum toxin vs. enteral neuromodulation in chronic refractory constipation. BMC Pediatr. 2023;23(1):448. DOI: 10.1186/s12887-023-04217-5
-62. Halleran DR, Lu PL, Ahmad H, et al. Anal sphincter botulinum toxin injection in children with functional anorectal and colonic disorders: a large institutional study and review of the literature focusing on complications. J Pediatr Surg. 2019;54(11):2305-2310. DOI: 10.1016/j.jpedsurg.2019.03.020
-63. Radwan AB, Gadallah MA, Shahawy MR, Albagdady AA, Talaat AA. Can botulinum toxin help in managing children with functional constipation and obstructed defecation?. J Pediatr Surg. 2021;56(4):750-753. DOI: 10.1016/j.jpedsurg.2020.06.044
-64. Engel BT. Clinical biofeedback: a behavioral analysis. Neurosci Biobehav Rev. 1981;5(3):397-400.
-65. Rao SS, Benninga MA, Bharucha AE, Chiarioni G, Di Lorenzo C, Whitehead WE. ANMS-ESNM position paper and consensus guidelines on biofeedback therapy for anorectal disorders. Neurogastroenterol Motil. 2015;27(5):594-609. DOI: 10.1111/nmo.12520
-66. Koh CE, Young CJ, Young JM, Solomon MJ. Systematic review of randomized controlled trials of the effectiveness of biofeedback for pelvic floor dysfunction. Br J Surg. 2008;95(9):1079-1087. DOI: 10.1002/bjs.6303
-67. Zubiri C, Miculan S, Zosi A, Nanfito GI, Guzman L, Borobia P, Bigliardi R, et al. Recto-anal biofeedback treatment and quality of life in children with myelomeningocele. Open J Epidemiol. 2019;9:202-212
-68. Cárcamo L, Aljaro S, Castro F, et al. Resultados a largo plazo de la rehabilitación del piso pélvico en pacientes con incontinencia fecal. Rev Cir. 2022;74(1):53-60. DOI: 10.35687/s2452-45492022001152
-69. Guerra-Mora J, Buenrostro-Acebes J, Erciga-Vergara N, et al. Efectividad del biofeedback en pacientes con incontinencia fecal. Rev Med Inst Mex Seguro Soc. 2015;53(4):472-475
-70. Sulkowski JP, Nacion KM, Deans KJ, et al. Sacral nerve stimulation: a promising therapy for fecal and urinary incontinence and constipation in children. J Pediatr Surg. 2015;50(10):1644-1647. DOI: 10.1016/j.jpedsurg.2015.03.043
-71. Dinning PG, Hunt LM, Arkwright JW, et al. Pancolonic motor response to subsensory and suprasensory sacral nerve stimulation in patients with slow-transit constipation. Br J Surg. 2012;99(7):1002-1010. DOI: 10.1002/bjs.8760
-72. Carrington EV, Evers J, Grossi U, et al. A systematic review of sacral nerve stimulation mechanisms in the treatment of fecal incontinence and constipation. Neurogastroenterol Motil. 2014;26(9):1222-1237. DOI: 10.1111/nmo.12388
-73. Dinning PG, Fuentealba SE, Kennedy ML, et al. Sacral nerve stimulation induces pan-colonic propagating pressure waves and increases defecation frequency in patients with slow-transit constipation. Colorectal Dis. 2007;9(2):123-132. DOI: 10.1111/j.1463-1318.2006.01096.x
-74. Knowles CH, Thin N, Gill K, et al. Prospective randomized double-blind study of temporary sacral nerve stimulation in patients with rectal evacuatory dysfunction and rectal hyposensitivity. Ann Surg. 2012;255(4):643-649. DOI: 10.1097/SLA.0b013e318247d49f
-75. Sacral nerve stimulation for the treatment of urinary and fecal incontinence. Available from: https://www.cms.gov/medicare-coverage-database/view/lcd.aspx?lcdid=39543&ver=5
-76. Humphreys MR, Vandersteen DR, Slezak JM, et al. Preliminary results of sacral neuromodulation in 23 children. J Urol. 2006;176(5):2227-2231. DOI: 10.1016/j.juro.2006.07.013
-77. van der Wilt AA, van Wunnik BP, Sturkenboom R, et al. Sacral neuromodulation in children and adolescents with chronic constipation refractory to conservative treatment. Int J Colorectal Dis. 2016;31(8):1459-1466. DOI: 10.1007/s00384-016-2604-8
-78. Dwyer ME, Vandersteen DR, Hollatz P, et al. Sacral neuromodulation for the dysfunctional elimination syndrome: a 10-year single-center experience with 105 consecutive children. Urology. 2014;84(4):911-917. DOI: 10.1016/j.urology.2014.03.059
-79. van Wunnik BP, Peeters B, Govaert B, et al. Sacral neuromodulation therapy: a promising treatment for adolescents with refractory functional constipation. Dis Colon Rectum. 2012;55(3):278-285. DOI: 10.1097/DCR.0b013e3182405c61
-80. Roth TJ, Vandersteen DR, Hollatz P, et al. Sacral neuromodulation for the dysfunctional elimination syndrome: a single-center experience with 20 children. J Urol. 2008;180(1):306-311. DOI: 10.1016/j.juro.2008.03.033
-81. Vriesman MH, Wang L, Park C, et al. Comparison of antegrade continence enema treatment and sacral nerve stimulation for children with severe functional constipation and fecal incontinence. Neurogastroenterol Motil. 2020;32(8):e13809. DOI: 10.1111/nmo.13809
-82. Lu PL, Asti L, Lodwick DL, et al. Sacral nerve stimulation allows for decreased antegrade continence enema use in children with severe constipation. J Pediatr Surg. 2017;52(4):558-562. DOI: 10.1016/j.jpedsurg.2016.11.003
-83. Park C, Wang L, Koppen I, et al. Sacral nerve stimulation leads to long-term improvement in fecal incontinence and quality of life for children with functional and organic defecation disorders. Neurogastroenterol Motil. 2024;36(9):e14865. DOI: 10.1111/nmo.14865
-84. Lu PL, Koppen IJN, Orsagh-Yentis DK, et al. Sacral nerve stimulation for constipation and fecal incontinence in children: long-term outcomes, patient benefit, and parent satisfaction. Neurogastroenterol Motil. 2018;30(2):e13184. DOI: 10.1111/nmo.13184
-85. Collins B, Norton C, Maeda Y. Percutaneous tibial nerve stimulation for slow transit constipation: a pilot study. Colorectal Dis. 2012;14(4):e165-e170. DOI: 10.1111/j.1463-1318.2011.02820.x
-86. Iqbal F, Collins B, Thomas GP, et al. Bilateral transcutaneous tibial nerve stimulation for chronic constipation. Colorectal Dis. 2016;18(2):173-178. DOI: 10.1111/codi.13105
-87. Rego RMP, Machado NC, Carvalho MA, et al. Transcutaneous posterior tibial nerve stimulation in children and adolescents with functional constipation: a protocol for an interventional study. Medicine (Baltimore). 2019;98(45):e17755. DOI: 10.1097/MD.0000000000017755
-88. Velasco-Benitez C, Villamarin E, Mendez M, et al. Efficacy of transcutaneous posterior tibial nerve stimulation in functional constipation. Eur J Pediatr. 2023;182(3):1309-1315. DOI: 10.1007/s00431-022-04798-w
-89. Yu ZT, Song JM, Qiao L, et al. A randomized, double-blind, controlled trial of percutaneous tibial nerve stimulation with pelvic floor exercises in the treatment of childhood constipation. Am J Gastroenterol. 2023;118(3):553-560. DOI: 10.14309/ajg.0000000000002188
-90. Bigliardi R, Riera Canales C, Oviedo A, et al. Successful use of transcutaneous parasacral stimulation in a patient with myelomeningocele and fecal incontinence: a case report. JPGN Rep. 2024;1-4. DOI: 10.1002/jpr3.12152
-91. Moore JS, Gibson PR, Burgell RE. Neuromodulation via interferential electrical stimulation as a novel therapy in gastrointestinal motility disorders. J Neurogastroenterol Motil. 2018;24(1):19-29. DOI: 10.5056/jnm17071
-92. Ng RT, Lee WS, Ang HL, et al. Transcutaneous electrical stimulation (TES) for treatment of constipation in children. Cochrane Database Syst Rev. 2016;11(11):CD010873. DOI: 10.1002/14651858.CD010873.pub4
-93. Chase J, Robertson VJ, Southwell B, et al. Pilot study using transcutaneous electrical stimulation (interferential current) to treat chronic treatment-resistant constipation and soiling in children. J Gastroenterol Hepatol. 2005;20(7):1054-1061. DOI: 10.1111/j.1440-1746.2005.03863.x
-94. Yik YI, Ismail KA, Hutson JM, et al. Home transcutaneous electrical stimulation to treat children with slow-transit constipation. J Pediatr Surg. 2012;47(6):1285-1290. DOI: 10.1016/j.jpedsurg.2012.03.037
-95. Clarke MC, Chase JW, Gibb S, et al. Decreased colonic transit time after transcutaneous interferential electrical stimulation in children with slow transit constipation. J Pediatr Surg. 2009;44(2):408-412. DOI: 10.1016/j.jpedsurg.2008.10.100
-96. Ismail KA, Chase J, Gibb S, et al. Daily transabdominal electrical stimulation at home increased defecation in children with slow-transit constipation: a pilot study. J Pediatr Surg. 2009;44(12):2388-2392. DOI: 10.1016/j.jpedsurg.2009.07.063
-97. Yik YI, Hutson J, Southwell B. Home-based transabdominal interferential electrical stimulation for six months improves paediatric slow transit constipation. Neuromodulation. 2018;21(7):676-681. DOI: 10.1111/ner.12734
-98. Besendörfer M, Kohl M, Schellerer V, et al. A pilot study of non-invasive sacral nerve stimulation in treatment of constipation in childhood and adolescence. Front Pediatr. 2020;8:169. DOI: 10.3389/fped.2020.00169
-99. Levitt MA, Pena A. Surgery and constipation: when, how, yes, or no?. J Pediatr Gastroenterol Nutr. 2005;41(Suppl 1):S58-S60
-100. Levitt MA, Martin CA, Falcone RA Jr, et al. Transanal rectosigmoid resection for severe intractable idiopathic constipation. J Pediatr Surg. 2009;44(6):1285-1290. DOI: 10.1016/j.jpedsurg.2009.02.049
-101. Levitt MA, Mathis KL, Pemberton JH. Surgical treatment for constipation in children and adults. Best Pract Res Clin Gastroenterol. 2011;25(1):167-179. DOI: 10.1016/j.bpg.2010.12.007
-102. Vilanova-Sanchez A, Levitt MA. Surgical interventions for functional constipation: an update. Eur J Pediatr Surg. 2020;30(5):413-419. DOI: 10.1055/s-0040-1716729
-103. Vriesman MH, Koppen IJN, Camilleri M, Di Lorenzo C, Benninga MA. Management of functional constipation in children and adults. Nat Rev Gastroenterol Hepatol. 2020;17(1):21-39. DOI: 10.1038/s41575-019-0222-y
-104. Yan Y, Sharma A, Herekar AA, Jimenez E, Hudgi AR, Gu QG, Rao SSC. Translumbosacral anorectal magnetic stimulation test for fecal incontinence. Dis Colon Rectum. 2022;65(1):83-92. DOI: 10.1097/DCR.0000000000002152
-105. Rao SSC, Xiang X, Sharma A, et al. Translumbosacral neuromodulation therapy for fecal incontinence: a randomized frequency response trial. Am J Gastroenterol. 2021;116(1):162-170. DOI: 10.14309/ajg.0000000000000766
-106. Rao SSC, Lembo A, Chey WD, et al. Effects of the vibrating capsule on colonic circadian rhythm and bowel symptoms in chronic idiopathic constipation. Neurogastroenterol Motil. 2020;32(11):e13890. DOI: 10.1111/nmo.13890
-107. Zhu JH, Qian YY, Pan J, et al. Efficacy and safety of vibrating capsule for functional constipation (VICONS): a randomized, double-blind, placebo-controlled, multicenter trial. EClinicalMedicine. 2022;47:101407. DOI: 10.1016/j.eclinm.2022.101407
-108. Mahler T. Education and hypnosis for treatment of functional gastrointestinal disorders (FGIDs) in pediatrics. Am J Clin Hypn. 2015;58(1):115-128. DOI: 10.1080/00029157.2015.1033676
-109. Palsson OS. Hypnosis treatment of gastrointestinal disorders: a comprehensive review of the empirical evidence. Am J Clin Hypn. 2015;58(2):134-158. DOI: 10.1080/00029157.2015.1039114
-110. Mayer EA, Tillisch K. The brain-gut axis in abdominal pain syndromes. Annu Rev Med. 2011;62:381-396. DOI: 10.1146/annurev-med-012309-103958
-111. Reed-Knight B, Claar RL, Schurman JV, van Tilburg MA. Implementing psychological therapies for functional GI disorders in children and adults. Expert Rev Gastroenterol Hepatol. 2016;10(9):981-984. DOI: 10.1080/17474124.2016.1207524
-112. van Ginkel R, Reitsma JB, Buller HA, et al. Childhood constipation: longitudinal follow-up beyond puberty. Gastroenterology. 2003;125(2):357-363
-113. Michaud L, Lamblin MD, Mairesse S, Turck D, Gottrand F. Outcome of functional constipation in childhood: a 10-year follow-up study. Clin Pediatr (Phila). 2009;48(1):26-31. DOI: 10.1177/0009922808320599
-114. Bongers ME, van Wijk MP, Reitsma JB, Benninga MA. Long-term prognosis for childhood constipation: clinical outcomes in adulthood. Pediatrics. 2010;126(1):e156-e162. DOI: 10.1542/peds.2009-1009
-115. DiMatteo MR, Giordani PJ, Lepper HS, Croghan TW. Patient adherence and medical treatment outcomes: a meta-analysis. Med Care. 2002;40(9):794-811
-116. Taddeo D, Egedy M, Frappier JY. Adherence to treatment in adolescents. Paediatr Child Health. 2008;13(1):19-24. DOI: 10.1093/pch/13.1.19
-117. Milgrom H, Bender B, Ackerson L, Bowry P, Smith B, Rand C. Noncompliance and treatment failure in children with asthma. J Allergy Clin Immunol. 1996;98(6 Pt 1):1051-1057. DOI: 10.1016/S0091-6749(96)80190-4
-118. Koppen IJN, van Wassenaer EA, Barendsen RW, Brand PL, Benninga MA. Adherence to polyethylene glycol treatment in children with functional constipation is associated with parental illness perceptions, satisfaction with treatment, and perceived treatment convenience. J Pediatr. 2018;199:132-139.e1. DOI: 10.1016/j.jpeds.2018.03.066
Published
How to Cite
Issue
Section
License
Copyright (c) 2026 Cecilia Zubiri, Anabella Zosi, Román Bigliardi

This work is licensed under a Creative Commons Attribution-NonCommercial-ShareAlike 4.0 International License.












