Semaglutide and Metabolic Dysfunction–Associated Fatty Liver Disease: A Systematic Review
DOI:
https://doi.org/10.52787/agl.v56i1.591Keywords:
Metabolic dysfunction-associated fatty liver disease, metabolic dysfunction-associated steatohepatitis, semaglutideAbstract
Introduction. Metabolic dysfunction–associated fatty liver disease (MAFLD) and metabolic dysfunction–associated steatohepatitis (MASH) are highly prevalent conditions and represent a major public health problem.
Objective. To conduct an updated systematic review of randomized controlled trials evaluating the impact of semaglutide on hepatic outcomes in patients with MAFLD/MASH.
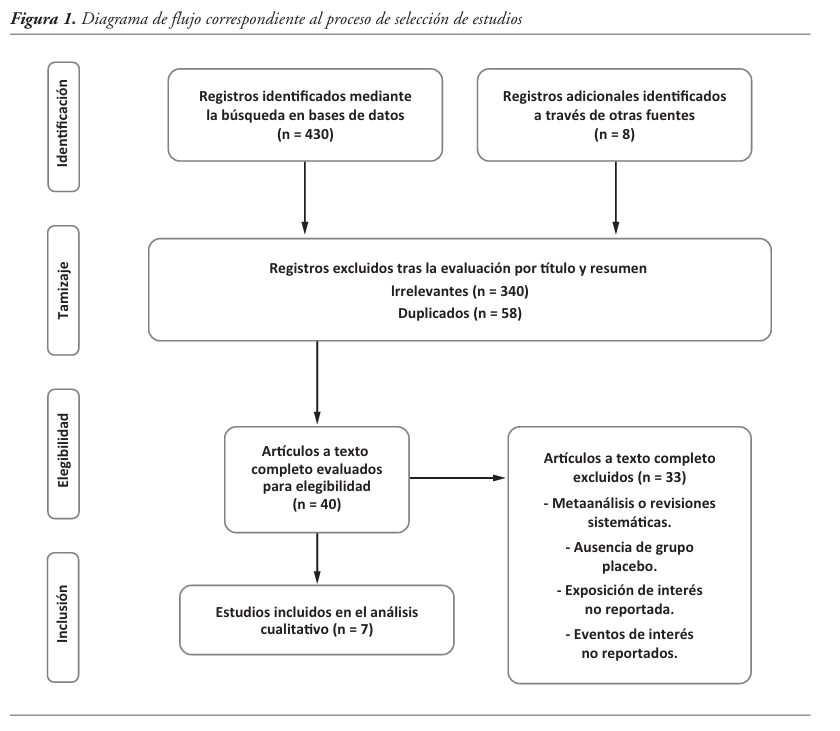
Materials and methods. A systematic literature search was performed in accordance with PRISMA guidelines. Randomized, placebo-controlled clinical trials assessing the effect of semaglutide on hepatic outcomes in patients with MAFLD/MASH were included, using histological parameters and/or imaging methods.
Results. The analyzed studies suggest a benefit of semaglutide compared with placebo in hepatic outcomes. Regarding the outcome of MASH resolution without worsening of fibrosis, consistent results were observed among studies that included liver biopsy. However, when evaluating fibrosis improvement without worsening of MASH, the results were heterogeneous. Imaging studies demonstrated a consistent benefit of semaglutide on hepatic steatosis. Nevertheless, no significant changes in liver stiffness were demonstrated. Differences in sample size, follow-up duration, and analyzed populations may explain these discrepancies.
Conclusion. The use of semaglutide in patients with MAFLD/MASH was associated with improvements in variables related to hepatic steatosis and, to a lesser extent, with changes in fibrosis markers. These findings should be confirmed in larger studies with longer follow-up periods.
References
-1. Younossi ZM, Kalligeros M, Henry L. Epidemiology of metabolic dysfunction-associated steatotic liver disease. Clin Mol Hepatol. 2025;31(Suppl):S32-S50. doi:10.3350/cmh.2024.0431.
-2. Basil B, Myke-Mbata BK, Eze OE, Akubue AU. From adiposity to steatosis: metabolic dysfunction-associated steatotic liver disease, a hepatic expression of metabolic syndrome - current insights and future directions. Clin Diabetes Endocrinol. 2024;10(1):39. doi:10.1186/s40842-024-00187-4.
-3. Quek J, Chan KE, Wong ZY, Tan C, Tan B, Lim WH, et al. Global prevalence of non-alcoholic fatty liver disease and non-alcoholic steatohepatitis in the overweight and obese population: a systematic review and meta-analysis. Lancet Gastroenterol Hepatol. 2023;8:20-30. doi:10.1016/S2468-1253(22)00317-X.
-4. Cho EE, Ang CZ, Quek J, Fu CE, Lim LKE, Heng ZEQ, et al. Global prevalence of non-alcoholic fatty liver disease in type 2 diabetes mellitus: an updated systematic review and meta-analysis. Gut. 2023;72(11):2138-2148. doi:10.1136/gutjnl-2023-330110.
-5. Marso SP, Bain SC, Consoli A, Eliaschewitz FG, Jódar E, Leiter LA, et al. Semaglutide and cardiovascular outcomes in patients with type 2 diabetes. N Engl J Med. 2016;375:1834-1844. doi:10.1056/NEJMoa1607141.
-6. Gouveri E, Popovic DS, Papanas N. Potential new therapeutic implications of semaglutide: new colours of the rainbow? Diabetes Ther. 2024;15(1):13-18. doi:10.1007/s13300-02301506-1.
-7. Amaro A, Sugimoto D, Wharton S. Efficacy and safety of semaglutide for weight management: evidence from the STEP program. Postgrad Med. 2022;134(sup1):5-17. doi:10.1080/00325481.2022.2147326.
-8. Lincoff AM, Brown-Frandsen K, Colhoun HM, Deanfield J, Emerson SS, Esbjerg S, et al. Semaglutide and cardiovascular outcomes in obesity without diabetes. N Engl J Med. 2023;389(24):2221-2232. doi:10.1056/NEJMoa2307563.
-9. Targher G, Byrne CD, Tilg H. MASLD: a systemic metabolic disorder with cardiovascular and malignant complications. Gut. 2024;73(4):691-702. doi:10.1136/gutjnl-2023-330595.
-10. Newsome PN, Buchholtz K, Cusi K, Linder M, Okanoue T, Ratziu V, et al. A placebo-controlled trial of subcutaneous semaglutide in nonalcoholic steatohepatitis. N Engl J Med. 2021;384(12):1113-1124. doi:10.1056/NEJMoa2028395.
-11. Flint A, Andersen G, Hockings P, Johansson L, Morsing A, Palle MS, et al. Randomised clinical trial: semaglutide versus placebo reduced liver steatosis but not liver stiffness in subjects with non-alcoholic fatty liver disease assessed by magnetic resonance imaging. Aliment Pharmacol Ther. 2021;54(9):1150-1161. doi:10.1111/apt.16608.
-12. Loomba R, Abdelmalek MF, Armstrong MJ, Jara M, Kjær MS, Krarup N, et al. Semaglutide 2.4 mg once weekly in patients with non-alcoholic steatohepatitis-related cirrhosis: a randomised, placebo-controlled phase 2 trial. Lancet Gastroenterol Hepatol. 2023;8(6):511-522. doi:10.1016/S2468-1253(23)00068-7.
-13. Ratziu V, Francque S, Behling CA, Cejvanovic V, Cortez-Pinto H, Iyer JS, et al. Artificial intelligence scoring of liver biopsies in a phase II trial of semaglutide in nonalcoholic steatohepatitis. Hepatology. 2024;80(1):173-185. doi:10.1097/HEP.0000000000000723.
-14. Schattenberg J, Grønbæk H, Kliers I, Ladelund S, Long MT, Nygård SB, et al. Proteomic signatures reflect effects of semaglutide treatment for MASH. JHEP Rep. 2025;7(10):101521. doi:10.1016/j.jhepr.2025.101521.
-15. Sanyal AJ, Newsome PN, Kliers I, Østergaard LH, Long MT, Kjær MS, et al. Phase 3 trial of semaglutide in metabolic dysfunction–associated steatohepatitis. N Engl J Med. 2025;392(21):2089-2099. doi:10.1056/NEJMoa241325.
-16. Golub IS, Manubolu VS, Aldana-Bitar J, Dahal S, Verghese D, Alalawi L, et al. The impact of semaglutide on liver fat assessed by serial cardiac CT scans in patients with type 2 diabetes: results from STOP trial. Nutr Metab Cardiovasc Dis. 2025;35(9):104036. doi:10.1016/j.numecd.2025.104036.
-17. Bandyopadhyay S, Das S, Samajdar SS, Joshi SR. Role of semaglutide in the treatment of nonalcoholic fatty liver disease or non-alcoholic steatohepatitis: a systematic review and meta-analysis. Diabetes Metab Syndr. 2023;17(10):102849. doi:10.1016/j.dsx.2023.102849.
-18. Mantovani A, Morandin R, Fiorio V, Lando MG, Stefan N, Tilg H, et al. Glucagon-like peptide-1 receptor agonists improve MASH and liver fibrosis: a meta-analysis of randomised controlled trials. Liver Int. 2025;45(9):e70256. doi:10.1111/liv.70256.
-19. Wang Y, Zhou Y, Wang Z, Ni Y, Prud'homme GJ, Wang Q. Efficacy of GLP-1-based therapies on metabolic dysfunction-associated steatotic liver disease and metabolic dysfunction-associated steatohepatitis: a systematic review and meta-analysis. J Clin Endocrinol Metab. 2025;110(10):2964-2979. doi:10.1210/clinem/dgaf336.
-20. Fang L, Li J, Zeng H, Liu J. Effects of GLP-1 receptor agonists on the degree of liver fibrosis and CRP in non-alcoholic fatty liver disease and non-alcoholic steatohepatitis: a systematic review and meta-analysis. Prim Care Diabetes. 2024;18(3):268-276. doi:10.1016/j.pcd.2024.03.005.
-21. Arya S, Kaji AH, Boermeester MA. PRISMA reporting guidelines for meta-analyses and systematic reviews. JAMA Surg. 2021;156:789-790. doi:10.1001/jamasurg.2021.0546.
-22. Sterne JAC, Savovic J, Page MJ, Elbers RG, Blencowe NS, Boutron I, et al. RoB 2: a revised tool for assessing risk of bias in randomised trials. BMJ. 2019;366:l4898. doi:10.1136/bmj.l4898.
-23. Alfawaz S, Burzangi A, Esmat A. Mechanisms of non-alcoholic fatty liver disease and beneficial effects of semaglutide: a review. Cureus. 2024;16(8):e67080. doi:10.7759/cureus.67080.
-24. Genua I, Cusi K. Approaches to nonalcoholic fatty liver disease: current and future therapies. Diabetes Spectr. 2024;37(1):48-58. doi:10.2337/dsi23-0012.
-25. Nogueira JP, Cusi K. Role of insulin resistance in the development of nonalcoholic fatty liver disease in people with type 2 diabetes: from bench to patient care. Diabetes Spectr. 2024;37(1):20-28. doi:10.2337/dsi23-0013.
-26. Vergès B, Duvillard L, Pais de Barros JP, Bouillet B, Baillot-Rudoni S, Rouland A, et al. Liraglutide increases the catabolism of apolipoprotein B100-containing lipoproteins in patients with type 2 diabetes and reduces proprotein convertase subtilisin/kexin type 9 expression. Diabetes Care. 2021;44(4):1027-1037. doi:10.2337/dc20-1843.
-27. Patel Chavez C, Cusi K, Kadiyala S. The emerging role of glucagon-like peptide-1 receptor agonists for the management of NAFLD. J Clin Endocrinol Metab. 2022;107(1):29-38. doi:10.1210/clinem/dgab578.
-28. Al-Qaaneh AM, Qunaibi EA, Al-Fraihat NA, Abu-Aisheh BE, Rabea S, Al Aloul AA, et al. Real-world off-label use of semaglutide for weight reduction: user behavior, effectiveness, and satisfaction. Patient Prefer Adherence. 2025;19:3373-3385. doi:10.2147/PPA.S549716.
-29. Duan Y, Pan X, Luo J, Xiao X, Li J, Bestman L, et al. Association of inflammatory cytokines with non-alcoholic fatty liver disease. Front Immunol. 2022;13:880298. doi:10.3389/fimmu.2022.880298.
-30. Masson W, Lobo M, Nogueira JP, Rodriguez-Granillo AM, Barbagelata LE, Siniawski D. Anti-inflammatory effect of semaglutide: updated systematic review and meta-analysis. Front Cardiovasc Med. 2024;11:1379189. doi:10.3389/fcvm.2024.1379189.
-31. Yang XD, Yang YY. Clinical pharmacokinetics of semaglutide: a systematic review. Drug Des Devel Ther. 2024;18:2555-2570. doi:10.2147/DDDT.S470826.
-32. Tang A, Desai A, Hamilton G, Wolfson T, Gamst AC, Lam J, et al. MRI and MRE for non-invasive quantitative assessment of hepatic steatosis and fibrosis in NAFLD and NASH: clinical trials to clinical practice. Nat Rev Gastroenterol Hepatol. 2022;19(5):341-359. doi:10.1016/j.jhep.2016.06.005.
-33. Arai T, Atsukawa M, Tsubota A, Ono H, Kawano T, Yoshida Y, et al. Efficacy and safety of oral semaglutide in patients with non-alcoholic fatty liver disease complicated by type 2 diabetes mellitus: a pilot study. JGH Open. 2022;6(7):503-511. doi:10.1002/jgh3.12780.
-34. Volpe S, Lisco G, Fanelli M, Racaniello D, Colaianni V, Triggiani D, et al. Once-weekly subcutaneous semaglutide improves fatty liver disease in patients with type 2 diabetes: a 52-week prospective real-life study. Nutrients. 2022;14(21):4673. doi:10.3390/nu14214673.
-35. Carretero-Gómez J, Carrasco-Sánchez FJ, Fernández-Rodríguez JM, Casado-Escribano P, Miramontes-González JP, Seguí-Ripoll JM, et al. Effect of semaglutide on fatty liver disease biomarkers in patients with diabetes and obesity. Rev Clin Esp (Barc). 2023;223(3):134-143. doi:10.1016/j.rceng.2022.12.001.
-36. Alkhouri N, Herring R, Kabler H, Kayali Z, Hassanein T, Kohli A, et al. Safety and efficacy of combination therapy with semaglutide, cilofexor and firsocostat in patients with non-alcoholic steatohepatitis: a randomised, open-label phase II trial. J Hepatol. 2022;77(3):607-618. doi:10.1016/j.jhep.2022.04.003.
-37. Romero-Gómez M, Lawitz E, Shankar RR, Chaudhri E, Liu J, Lam RLH, et al. A phase IIa active-comparator-controlled study to evaluate the efficacy and safety of efinopegdutide in patients with non-alcoholic fatty liver disease. J Hepatol. 2023;79(4):888-897. doi:10.1016/j.jhep.2023.05.013.
Published
How to Cite
Issue
Section
License
Copyright (c) 2026 Walter Masson, Carla Solís, Juan Patricio Nogueira

This work is licensed under a Creative Commons Attribution-NonCommercial-ShareAlike 4.0 International License.












